Download a printable version of this document here

Adsorption and absorption mean quite different things.
Absorption is where a liquid is soaked up into something like a sponge, cloth or filter paper. The liquid is completely absorbed into the absorbent material.
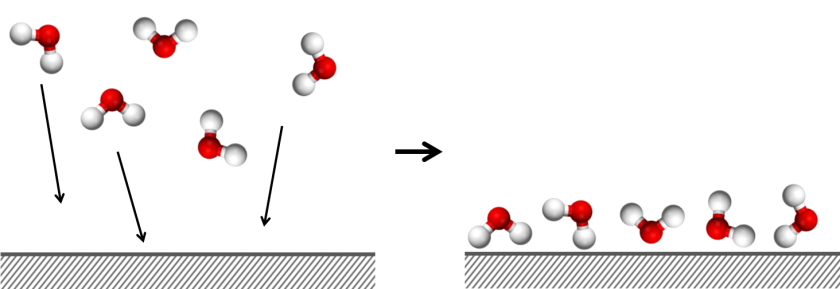
Adsorption refers to individual molecules, atoms or ions gathering on surfaces. The surface of a material is made up of atoms and bonds that are exposed to the air. For example, the surface of a piece of glass will be covered in silicon and oxygen atoms. Molecules or ions can interact with this surface via intermolecular interactions. This allows them to ‘stick’, or adsorb, to the surface. If a material has a very high surface area, then lots of molecules can stick to the surface.

This work is licensed under a Creative Commons Attribution 4.0 International License.