Background
Most of the elements in the periodic table are metals. Metals have fascinating properties and are essential for our buildings, technological devices and even in healthcare. Some metals are essential for our bodies to function properly, e.g. iron in haemoglobin to transport oxygen around our body.
Exercise 1: Choose a metallic element from the periodic table. Try and pick something you are unfamiliar with. Look it up on the internet and find out some of the applications it is used in.
However, if certain metals get into our water supply they could cause problems. Some metals are toxic and can cause damage to our bodies.
 Metals can enter waterways in many different ways. Some leach (dissolve) into ground water from the minerals that exist in the Earth’s crust. An example of this is arsenic, which is present in the groundwater in many countries and has caused many illnesses and deaths. Metals can also enter waterways from industry or from consumers. This could be through lack of regulation, poor implementation of regulation or from accidents. For example, the Fukushima Daiichi Nuclear Power Station in Japan was damaged by the 2011 earthquake and tsunami. Following work to cool the reactors with seawater, water in the harbour and groundwater was contaminated with radioactive elements.
Metals can enter waterways in many different ways. Some leach (dissolve) into ground water from the minerals that exist in the Earth’s crust. An example of this is arsenic, which is present in the groundwater in many countries and has caused many illnesses and deaths. Metals can also enter waterways from industry or from consumers. This could be through lack of regulation, poor implementation of regulation or from accidents. For example, the Fukushima Daiichi Nuclear Power Station in Japan was damaged by the 2011 earthquake and tsunami. Following work to cool the reactors with seawater, water in the harbour and groundwater was contaminated with radioactive elements.
One way to remove metals from water is to use adsorbents. These can include activated carbons or zeolites.
Zeolites to trap metals
Zeolites are a type of crystalline solid material that occur naturally in the Earth’s crust. They can also be synthesized in the laboratory. Zeolites are made up of silicon and aluminium atoms, which are connected together via their oxygen atoms. These link into a three-dimensional crystalline structure with molecule-sized tunnels and cavities, to build a cage-like structure:
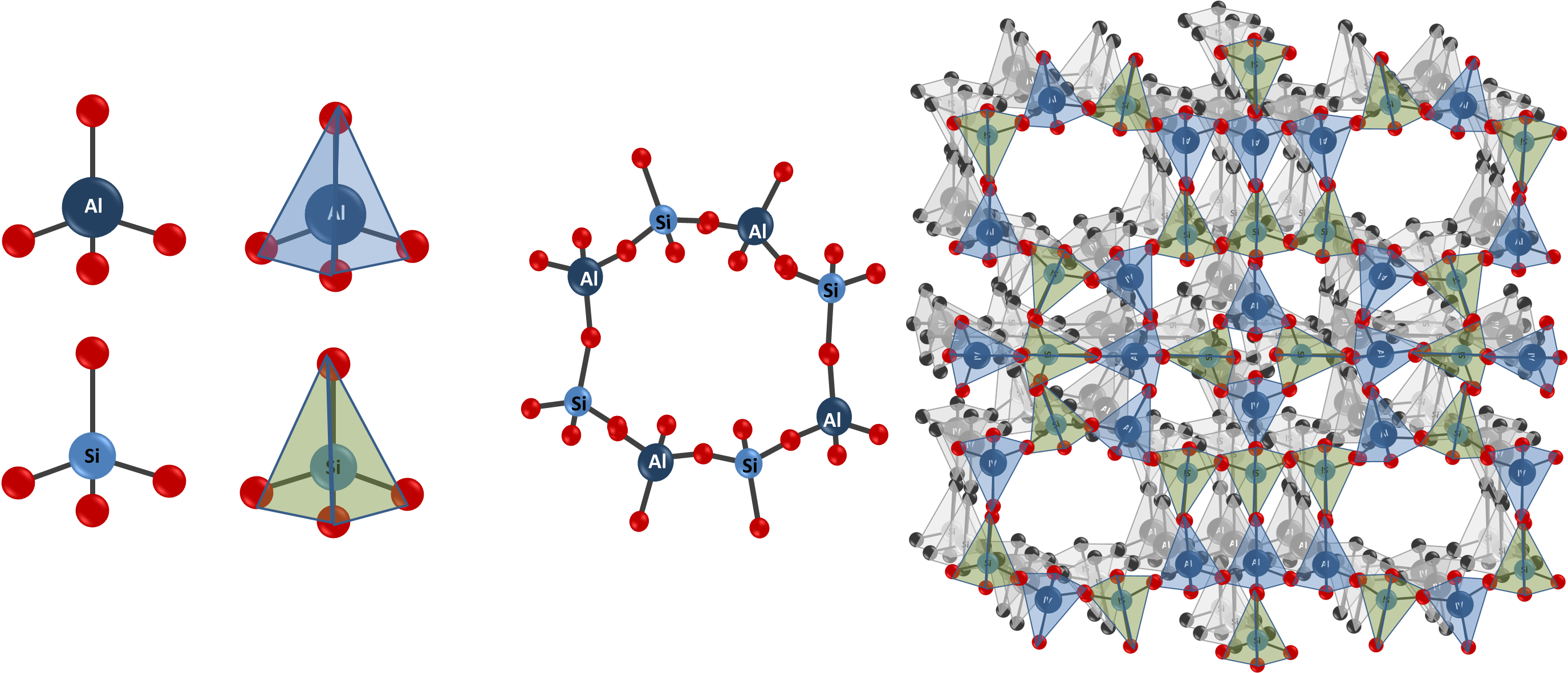
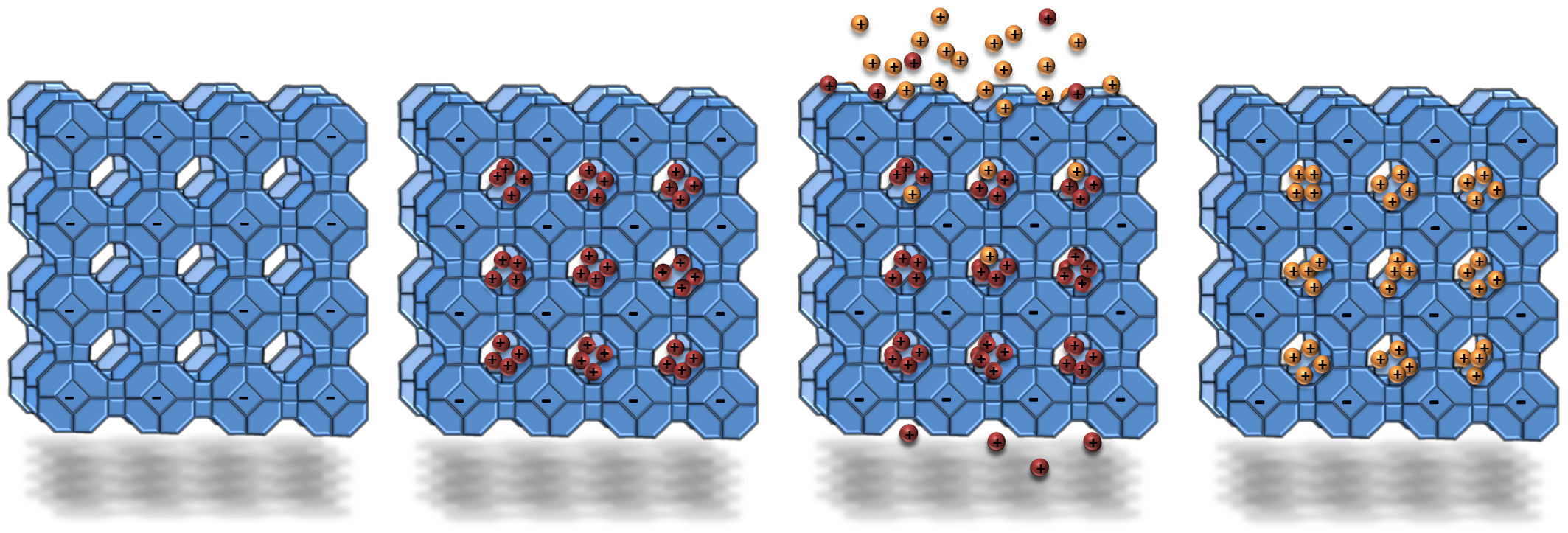
The 3D framework of zeolites can behave as ‘cages’ that can be filled with ions. They are microporous, which means that they contain pores that are less than 2 nm in diameter. This means that they can adsorb molecules and ions that are smaller than the size of the pore openings. The cage framework has an overall negative charge. The negative charge of the framework is balanced by positively-charged ions in the holes in the cage.

Exchanging the ions in zeolites
The cations that sit within a zeolite framework are mobile and can be exchanged for other cations. The schematic below shows a blue zeolite (aluminosilicate) framework that is negatively charged. The negative charges are balanced by positively charged ions (red) filling the holes. The red cations can be exchanged for other cations (yellow).
Ion exchange works particularly well if the second type of cation has a higher charge. It can also work if the second cation has a better size-match for the pore in the zeolite.
Ion exchange in zeolites is used widely in industry. For example, an industry may use zeolites to remove toxic metals from their waste water streams. This ensures that the water going back into waterways is safe. Zeolites are also in laundry detergents to reduce the formation of soap scum. Find out how ion exchange works in laundry detergents here.
Zeolites and other cage-like materials can be used to trap all sorts of ions, including radioactive ions; researchers at the University of Birmingham study the way in which zeolites and other porous crystalline structures can exchange ions with radioactive ions, in order to clean up nuclear waste.
Zeolites don’t just trap ions. For example, they are also found in pet litter! The porous crystalline structure of the zeolites helps by trapping unwanted liquids and odour molecules. Zeolites are also used as catalysts, for example in the ‘cracking’ process of alkenes. Again, it’s the porous structure of zeolites that proves important. The many pores in a zeolite’s open structure are like millions of tiny test tubes where atoms and molecules become trapped and chemical reactions readily take place. Since the pores in a particular zeolite are of a fixed size and shape, zeolite catalysts can work selectively on certain molecules.
The experiment
You might consider combining this activity with one that looks at removal of organic pollutants using activated carbons.
AIM
To remove metal ions from a solution using a zeolite
YOU WILL NEED:
- glass conical flasks
- 2% w/v copper(II) sulfate (aq) solution
- 0.1% w/v citric acid (aq) solution
- universal indicator
- Zeolite A powder (4 Å molecular sieve powder)
- measuring cylinder
- stirring rod
- plastic Pasteur pipette
- test tubes / vials
- spatula
- filter paper
- weighing boats
PROCEDURE
Measure out 50 mL of copper sulfate solution into two glass conical flasks, labelled A and B. Keep flask A as a control solution. Add 1 g of zeolite powder to solution B, and swirl the solution, continually for 5 min. Leave the solution to settle and move on to part 2.
Carefully remove 2 mL of solution A and solution B (without disturbing the solid at the bottom) into separate test tubes / vials, and compare the colours. Finally, scoop out some of the solid from the bottom of solution B, onto some filter paper, and observe its colour.
QUESTIONS
- Look at solutions A and B. When copper(II) ions are present in water, they have a blue colour. Compare the colour of the solutions taken from A and B – what do you notice?
- Looking at the zeolite that has settled in solution B, what colour is this? What does this tell you about where the copper ions have gone from the solution?
- What ions have come out of the zeolite in this experiment.
- We have zeolites in the water filters in our dishwashers to ‘soften water’. As the hard water runs through the filter, Ca2+ and Mg 2+ ions exchange with Na+ ions in the filter. Why do you think we have to add dishwasher salt (NaCl) to the filter every so often?
- How do you think zeolites could be used to clean up nuclear waste in contaminated water? Once the zeolites have swapped all their Na+ ions with radioactive ions, what do you think is the main problem that we need to overcome?
EXTENSION ACTIVITY
Measure out 100 mL of citric acid solution into a beaker, and add 0.5 mL of universal indicator into the solution. Stir the solution, and note the colour and pH of this solution. Divide the solution into two glass conical flasks, labelled C and D. Keep flask C as a control solution, and add 1 g of zeolite powder to the solution, and swirl for 1 min. Note the colour change that you observe. Leave the solution to settle for 5 min.
QUESTIONS
- Looking at solutions C and D – what is the pH of solution C? What is the pH of solution D? pH is a measure of H+ concentration, as the pH gets lower, the concentration of H+ increases. Which solution contains more H+?
- Can you explain what has happened to the solution in terms of ion exchange with the zeolite?
In the research lab
Researchers at the University of Birmingham use zeolite-type structures of porous materials to exchange ions with radioactive ions that are found in nuclear waste. Researchers, including Dr Tzu-Yu Chen, in the group of Dr Joe Hriljac, have recently looked at the ion-exchange of radioactive isotopes of cesium using cage-type crystalline structures, similar to zeolites. Radioactive cesium is produced during the uranium fission in nuclear reactors, and is also the main medium-term health risk remaining from the Fukushima accident.
Ion exchange is performed by passing contaminated ground water, containing radioactive ions, through columns containing porous zeolite-type structures. Once used, and full of radioactive ions, these columns are then classified as high-level radioative waste, and need to be safely stored; the ions are mobile and the exchange is reversible – so once trapped inside the cage-structures – how do we stop them leaching out? Researchers in Dr Joe Hriljac’s lab investigate how the chemical structure of the cage-like materials can be changed using heat and pressure, to lock the ions in place permanently, using a process called ‘hot isostatic pressing’:
To find out more you can read an open-access paper by clicking here.
Page author: Nicola Rogers Simpson
Edited by Emily Hanover 07/07/2022
Images on this page: engine parts by Aaron Barnaby and water by mrjn Photography on Unsplash,

This work is licensed under a Creative Commons Attribution 4.0 International License.