 Background
Background
Thin layer chromatography (TLC) is used routinely in the laboratory to both monitor reactions and analyse the purity of samples. TLC is a type of adsorption chromatography, and the most common substrates used for the stationary phases in the lab, are silica (SiO2) and alumina (Al2O3). It is recommended that you read the page on adsorption chromatography before doing this experiment.
Chromatography has two main uses: it is either used to test how pure something is, or is used as a technique to purify something from a mixture.
Chromatography is vital for any chemical research, because a chemical reaction rarely gives us 100 % pure product; we usually get some side-products, and some unreacted starting material that we need to separate from our desired product. The Environmental Agency uses chromatography to test drinking water and to monitor air quality. Pharmaceutical companies use chromatography both to prepare large quantities of extremely pure materials, and also to analyze the purified compounds for trace contaminants. Chromatography is used for quality control in the food industry, by separating and analyzing additives and proteins, and is used for finding drug compounds in urine or other body fluids, for example when testing athletes for drug use.
You may already be familiar with paper chromatography, where the paper (cellulose) behaves as a backing plate, and water molecules that hydrogen-bond to the surface form the stationary phase. The solvent (often water or ethanol) forms the mobile phase and moves across the surface through capillary action.
Researchers in Chemistry use silica and alumina TLC regularly, to monitor how reactions are progressing, or to analyse fractions that come off columns during purification. This technique is used whilst synthesising new materials for a whole host of applications, including drug discovery, molecular sensors, and new fluorescent dyes.
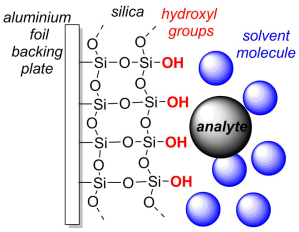
Silica TLC plates are usually supplied as silica powder supported on a plate of aluminium foil. (Large plates can be cut into small plates with scissors, typically 3 x 6 cm). The hydroxyl-terminated silica adsorbs analytes (the solute) to varying degrees, and an organic solvent is used as the mobile phase, the eluent. On a TLC plate the solvent percolates through the solid by capillary action from the bottom of the plate. To read more about running and visualising TLC plates click here to read the section on the adsorption chromatography page.
Experiment
Instructions for students, teachers and technicians are available to download at the bottom of the page. Please ensure you refer to the safety card.
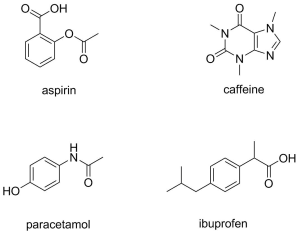
There are many drug options available on the market to buy over the counter for pain relief, many of which we recognise by their brand name, and often they contain combinations of active drug ingredients. The most common pain relief drugs include aspirin, paracetamol, and ibuprofen, and often these are combined with caffeine to increase the effectiveness of the pain relief.
This video gives a first-person view of this experiment being carried out:
AIM
To analyse the components of painkiller tablets by comparison to known standards
YOU WILL NEED:

- Fluorescent silica gel TLC plates
- 100 mL glass beaker and a small watch glass (or a wide jam jar with a screw lid)
- Spatula
- 6 glass vials or small beakers
- Filter paper
- Ethanol
- Ethyl acetate
- Micro-pipette spotters for TLC (these can be made with a Bunsen burner from capilliary tubes or glass pasteur pipettes)
- Caffeine tablet
- Paracetamol tablet
- Aspirin tablet
- Ibuprofen tablet
- mixed painkiller tablets X and Y
- UV TLC lamp (254 nm wavelength, short wave)
- Permanganate dip and tweezers
- Paper towels
PROCEDURE
Crush up 1 aspirin tablet with a spatula, and add 2-3 mL of ethanol, in a vial or small beaker, and label the beaker ‘ethanolic aspirin solution’ . Repeat this with a caffeine, paracetamol and ibuprofen tablet, to make up your 4 known standard solutions – if the drug is in a capsule, you can open the capsule and tip the powder into the ethanol. Stir the solutions for several minutes to try to dissolve as much as possible (you can shake them if they are in vials with lids). Finally, take a tablet/powder of 2 unknown mixtures of painkillers, and repeat the procedure above to make a solution labelled ‘ethanolic solution X’, and ‘ethanolic solution Y’.
Now you need to make yourself a TLC tank, with either a 100 mL beaker and a watch glass,or a wide jam jar with a screw-top lid.
Alternatively foil can be used to make a lid. Line the tank with filter paper (this helps to saturate the air in the tank with solvent), and pour a small amount (approximately 10 mL) of ethyl acetate into the tank – if you are doing this outside of a fume hood, do not leave the tank without a lid on. Allow the ethyl acetate to soak up into the filter paper (you can swirl the tank to do this), and either add or remove some ethyl acetate so that the solvent level is roughly 0.5 cm high.
To load your TLC plate, first draw a line on the plate in pencil (not ink as this will run with the solvent up the plate) 1.0 cm from the bottom of the plate; this marks the starting point. Mark 6 dashes along this line, in pencil, as markers for your samples: aspirin, paracetamol, caffeine, ibuprofen, X and Y. Be careful to mark the silica gently, without scoring it.

Then dip a micro-pipette into a solution of your sample (just take the liquid from your premade solutions, leave the solid to settle to the bottom), and make a tight spot on the plate by lightly touching the plate on the baseline at a marked point. Make sure you number/label these so that you know which lane is which drug. Now check the plate before you develop it under the UV TLC lamp, to make sure the sample is strong enough to see – you should be able to see a dark spot in each lane along the baseline. Make sure that the solvent has evaporated from the spots before you develop the plate. You can reuse the same micropipette, as long as you draw up some ethanol and spot it onto some paper towel, between samples.
Caffeine, paracetamol, aspirin, and ibuprofen all absorb UV light, and can be identified on the fluorescent silica plates under short wave (254 nm) UV radiation; when viewed under short wave UV the zinc sulfide in the silica plates fluoresces green, except where an eluted substance quenches this fluorescence – these stand out as dark spots on the plate. If the sample is too weak, allow the spot to dry and reapply. This can be done several times, but allow the spot to dry each time to keep the spot size small.
Place the loaded TLC plate into the tank carefully, making sure the baseline is at the bottom, the back of the plate leans against the tank wall at a slight angle, and the baseline is above the level of the eluent. Do not move the tank during the plate development. Place the lid (watch glass) on the tank and allow the eluent to rise up the plate, until it is about 1 cm from the top. Carefully remove the plate, mark the solvent front with a pencil, and allow the plate to dry (preferentially in a fume hood).
Look at the TLC plate under 254 nm UV light, and use a pencil to circle the dark spots that you can see on the plate. Measure the Rf (retention factor) of each of the standards, and draw a copy of your TLC. Dip your plate into a permanganate dip using tweezers, and put it onto a paper towel to dry, aluminium side down against the towel. Avoid getting the permanganate solution on your hands. Once the plate has dried, draw what you observe, and then determine by comparison which drugs are in X and Y.
If you are struggling to understand your TLC plate, take a look at our ‘TLC troubleshooting‘ page.
QUESTIONS
-
- What is the retention factor (Rf) for each of the 4 standard drugs, measured from the UV visulasation?
- Looking at the structures of the 4 different drugs below, what do they all have in common, and why do you think they absorb UV light?
- What are the stationary/mobile phases of this chromatographic technique?
- Why is it important to use a pencil, and not pen, for marking TLC plates?
- Which compounds stain immediately with the permanganate dip? Why do you think this is?
- What combination of drugs is in the unknown mixtures?
- When recording the Rf of a compound, what factors are important to know about the TLC run? What factors can change the Rf?
For A Level practical resources related to organic synthesis, take a look at the University of Birmingham page about making aspirin.
In the research lab
Below is a set of photographs taken whilst performing adsorption chromatography by Dr Nicola Rogers, whilst working as a research chemist at Durham University. Compounds were synthesised and purified by chromatography, in order to make new imaging agents that can help give contrast in MRI scans.
The photographed chromatography was used to make the lanthanide complexes published in the following research paper:

is work is licensed under a Creative Commons Attribution 4.0 International License.



