Background
Nanotechnology is becoming increasingly important in modern times and already has many uses in medical applications and technology.
Nanotechnology is the use and control of structures and materials that are on the nanoscale (between 1-100 nm). 1 nm is 1 billionth of a metre, i.e. 0.000 000 001 m. To put this in perspective, a human hair is typically 100,000 nm in diameter.
A nanomaterial is any material that has one of its dimensions on the nanoscale. This can include nanoparticles, which are 1-100 nm in diameter. But nanomaterials can also be very long in one direction. For example, DNA is only a few nanometres across, but can be several centimetres long. Nanomaterials can also include sheet-like materials like graphene.
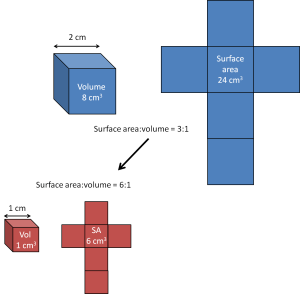
Nanomaterials are all around us and can occur naturally, for example in volcanic ash. Many, however, are designed and produced by scientists. Nanomaterials often behave quite differently to their bulk materials, merely due to the nanoscale dimensions of the material. Nanomaterials have very large surface areas, and are often more reactive. A term that is often used by scientists is that nanomaterials have a high ‘surface-to-volume ratio’.
Exercise 1: Surface-to-volume ratio worksheet.
Nanostructured surfaces
An important area of nanotechnology is creating surfaces with certain properties. Scientists often take inspiration from nature with this as some plants and animals have nanostructured surfaces. The following activity will look at how a nanostructured surface can make a material repel water, why it might want to do that and why we might want to copy that.
The experiment
AIM
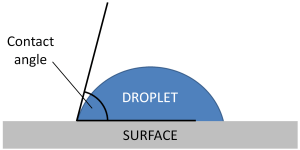
To investigate how different surfaces interact with a droplet of water and measure the contact angle of the droplet with each surface.
YOU WILL NEED
- a plastic Pasteur pipette
- 1-2 mL of tap water
- a square of a dried lotus leaf ~ 6 x 6 cm (these can be bought online or in large packs from many asian supermarkets.
- squares of other materials – plastic sheet, paper, kitchen foil
- a glass microscope slide
PROCEDURE
Firstly take a sheet of paper, and drop a droplet of water onto the paper. Look at the droplets side-on, and draw the shape that you see. Tilt the paper and see if the droplet likes to ‘stick’ to the paper. Repeat this with foil and plastic and the glass microscope slide. Finally, take a dried lotus leaf and try this. Here is a video of the lotus leaf in action!
Take a side-on picture of each droplet from the experiment above. Print your image and estimate the contact angle for each droplet. If you don’t have a camera, you could use the images below (click to access the full-size file). The angle only needs to be a rough estimate.





You can measure the contact angle by drawing one line along the surface and another tangent to the curve of the droplet where it touches the surface.
A small contact angle shows that the water sticks well to the surface. We say the surface is hydrophilic.
A large contact angle (>90°) means that the surface is hydrophobic.
If there is a very large contact angle (>150°), we call the surface superhydrophobic or ultrahydrophobic.
Use the table below to classify the type of surface on each of your 5 materials.
| Contact angle value | Type of surface |
| ~0° | Super-hydrophilic |
| <30° | Hydrophilic |
| 30-90° | Intermediate |
| 90-150° | Hydrophobic |
| >150° | Superhydrophobic |
EXTENSION ACTIVITY
If you are able to access some microscopes, you can look at the surface of the lotus leaf (see examples below). Compare them to the surfaces of other materials and perhaps other leaves?
Nanotechnology
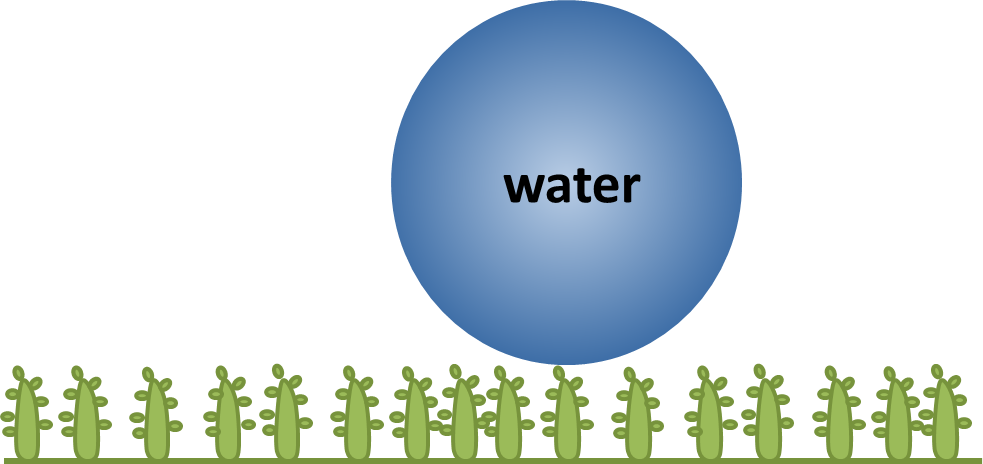
The lotus leaf is an example of a functional nanosurface that exists in nature. The surface of a lotus leaf is ‘superhydrophobic’; when water is dripped onto its surface it does not wet the leaf at all, but rolls off. Not only are the leaves extremely water-repellent, they are also ‘self-cleaning’, due to the surface structure of the leaves. The lotus leaf has a series of protrusions that are roughly 10 μm (1.0 x 10– 5 m) high covering its upper surface, these can be seen in the microscope images shown below:


This ‘rough’ surface structure with microscale bumps has a second smaller structure as each protrusion is itself covered in bumps of a hydrophobic, waxy material that are roughly 100 nm (1 x 10-7 m) in height. This means that water droplets sit lightly on the tips of hydrophobic protrusions as if on a bed of nails. This combined structure traps a layer of air in between the surface of the leaf and the water droplet. Hence, the water is not allowed to wet the surface and is easily displaced. This superhydrophobicity gives rise to the self-cleaning process on the surface because as the droplets travel along the leaves, they pick up any dirt or other matter they encounter along the way. This process keeps the lotus leaves dry, clean and free of pathogens such as bacteria and fungi.
In the research lab: self-cleaning and water-repellent surfaces

Chemists at the University of Durham have developed methods of creating similar surface-structures artificially, that can be sprayed onto devices and protect them from water damage. The invention has been commercialised by the British company, P2i, which is the world leader in liquid repellent nanocoating technology and was named ‘most innovative company in Europe’ in the 2012 International Business Awards. Customers include household names such as Nokia, Motorola and Alcatel (mobile phones), Timberland and Hi-Tec (footwear) and HLT (hearing aids).
An excellent article about superhydrophobic materials from ‘Chemistry World’ can be found here.
In the research lab: detecting prostate cancer
Researchers at the University of Birmingham, including Professor Paula Mendez in Chemical Engineering, and the late Professor John Fossey (School of Chemistry) create functional nanosurfaces to make sensors to detect diseases, such as prostate cancer. Sensors are one of the many areas of technology that rely on the development of new nanomaterials, and their manufacturing techniques. Information about our health can be extracted from biological material in our saliva, blood, urine and sweat, in order to detect the onset of disease. Many of these ‘disease markers’ include proteins and DNA, which are tens of nanometers in size, and hence interact selectively with materials on the nanoscale. The sooner a disease is diagnosed, the more likely it can managed or cured. To learn more about these sensors, take a look at the video below, published April 2016:

This work is licensed under a Creative Commons Attribution 4.0 International License.