
Natural dyes found in leaves
Natural dyes, also referred to as pigments, are organic compounds whose organic structure allows them to absorb light in the visible region of spectra. These compounds are responsible for the colour of all plants and trees. Looking at them from the perspective of a chemist, dyes are a diverse group of compounds, however they all have one attribute in common – high conjugation. Molecules of pigments are highly conjugated, meaning there is altering single and double bonds. This high conjugation allows them to absorb visible light and hence we perceive them as coloured.
In leaves we see Chlorophylls as the main dyes, however other dyes are present as well depending on the type and colour (e.g. green/red) of the leaf. The other most common dyes include Xanthophylls, Anthocyanins and Melanins.
Chlorophyll
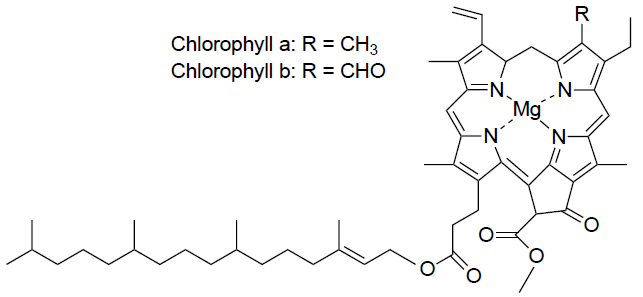
Chlorophylls are the most important dyes as they play a role in photosynthesis. As of today, seven types of chlorophylls have been detected – chlorophyll a, b, g, d, e, bacteriochlorophyll and bacterioviridin. The most significant ones of these are chlorophyll a and chlorophyll b since they contribute to photosynthesis.
These green pigments can be found in tissue of green plants. More precisely, they are located in chloroplast (an organelle in plant cell). The two types of chlorophylls have a slightly different colour and chemical structure. Which chlorophyll is the major one depends on whether the plant is a Heliophyte (Sunstroke plant) or a Sciophyte (prefers low light). Heliophytes have chlorophyll a as the major one while it is the other way around in Sciophytes.
In terms of colour, chlorophyll a is blue-green coloured while chlorophyll b is yellow-green. Looking at the chemical structure, the difference between the two types is in the functional group adjacent to the second pyrrole of the compound (see image below). Chlorophyll a has a methyl group adjacent to the pyrrole and chlorophyll b has an aldehyde. A characteristic to both is magnesium ion (Mg 2+) located in the centre of the heme structure.
Photosynthesis is a chemical reaction during which light energy is converted into chemical energy. Carbon dioxide (CO2) and water are transformed into glucose (C6H12O6) and oxygen. The reaction can be simplified as:
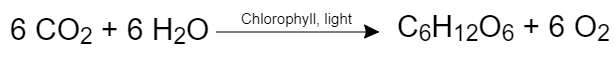
Sunlight and higher temperatures are needed for the production of Chlorophylls in leaves. As a consequence of temperatures dropping down and less of sunlight in autumn, the production of Chlorophylls in leaves reduces and the already produced Chlorophylls slowly begin to degrade. Other natural dyes (e.g. Xanthophylls) are also affected by these changes and begin to degrade, however they degrade at a slower rate than Chlorophylls and so become visible in leaves as they superimpose the green dyes.
Exercise: identifying common organic functional groups in chlorophyll a and b here.
Xanthophyll
Xanthophylls are part of a group of dyes called Carotenoids (also called Tetraterpenoids) which can be yellow, orange or red in colour. They can be found in leaves and fruits alongside with chlorophylls. One of the most common Xanthophylls is yellow-coloured Lutein (see image for structure). Carotenoids also play a role in photosynthesis. They protect the photosynthetic apparatus (photosystem) from a strong sunlight. Furthermore, Tetraterpenoids can absorb light of a different wavelength from Chlorophylls so a broader spectrum of light can be absorbed by a plant.
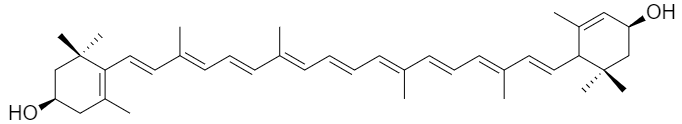
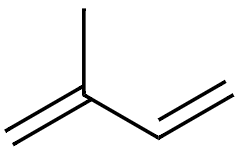
Looking at the chemical structure, Carotenoids consist of a certain number of isoprene groups (isoprene = 2-methyl-1, 3-butadiene). The main body of the structure is made up of eight isoprene units, hence the name tetraterpenoids. Xanthophylls are oxide derivatives of tetraterpenoids.
Anthocyanin
Anthocyanins are one of the most widespread Pyran dyes. They can be orange, red or even purple and blue coloured. Their role in leaves is to protect them from strong sunlight as well as attract pollinators with their bright colours.
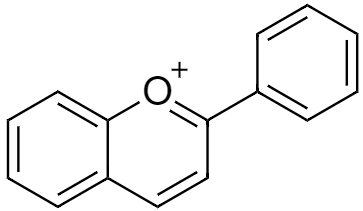
Considering chemical structure, Anthocyanins are made up of Flavylium cation and a number of hydroxyl groups (–OH) and saccharides bonded to the cation in different places depending on the type of anthocyanin. Some anthocyanins also have methoxide groups (–OCH3) attached to the cation. Saccharides (e.g. glucose, galactose) are bonded to the cation by the Glycosidic bond (–O–). An example of one Anthocyanin could be cyanidin-3-O-β-D-glucoside.
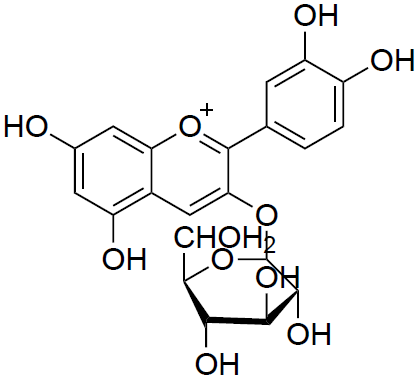
Anthocyanins are used as food colouring in wine, juice, yogurt, various desserts or ice cream. Considering their anti-inflammatory and anticarcinogenic properties, some are also used in pharmaceutical industry.
Melanin
Melanins are an example of Chinin dyes, one of the most widespread groups of dyes in nature. They can be different shades of yellow, brown or black. These dyes can be found in roots, tree bark and in autumn they are most apparent in leaves.
In terms of chemical structure, there is not a uniform structure like there is in Chlorophylls, Xanthophylls and Anthocyanins.
Melanins are used in food and pharmaceutical industries as well as cosmetics for their detoxifying and photoprotective (they can absorb UV light and hence protect) properties.
Laboratory techniques used for the experiment
In the experiment we will be using Paper Chromatography (PC) and Liquid-Liquid Extraction (LLE).
Chromatography
Chromatography is a separation method enabling scientist to separate individual compounds from a mixture. A Russian botanic Mikhail Tswett, who in 1901 worked on a research about natural dyes and used chromatography, is considered as the inventor of this separation technique. He named the method “chromatography” from the Greek words “chroma” (= colour) and “graphos” (= to write). Nowadays, there are several different methods of chromatography used in labs. The most common ones include Gas Chromatography (GC) and High Performance Liquid Chromatography (HPLC). More on chromatography can be found here.
In this experiment, we will be using Paper Chromatography (PC) which is similar to commonly used Thin Layer Chromatography (TLC).
Paper Chromatography (PC) is a simple method used for determining the composition of a sample. A sample is spot onto a filtration paper and the bottom of the paper is then dipped into an appropriate solvent (the sample spot must be above the level of the solvent). The solvent is carried across the paper and takes the sample with it. Individual compounds in the sample are carried with different speeds and so they separate.
There are different ways of developing the chromatogram which is illustrated in the following scheme:
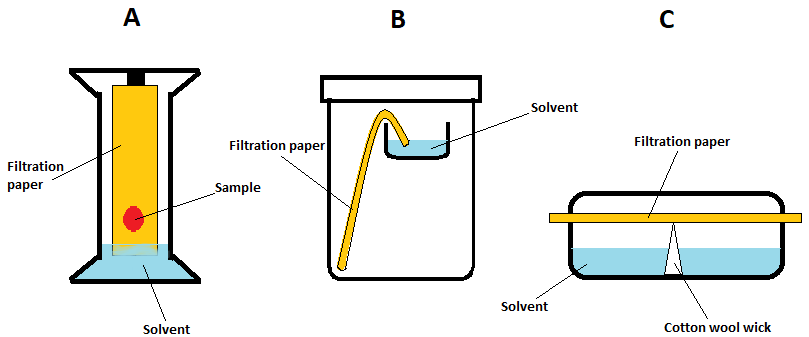
For the purpose of our experiment, we will be using method C.
Extraction
Extraction is a separation method used in chemical, petrochemical, pharmaceutical and food industries. For example, it is used during the manufacture of penicillin and to extract the active substance from plants.
Liquid-Liquid Extraction (LLE) is based on the principle of immiscibility of solvents. We add a second solvent to our sample already in a certain solvent. The second solvent has to be immiscible with the first solvent while the sample has to be miscible in both solvents. After the mixing, a two-phase mixture is obtained (liquid-liquid) and sample is transported between the two solvents. Upon equilibration, individual components of the sample are separated in the two immiscible solvents. They can be then separated from each other based on their density.
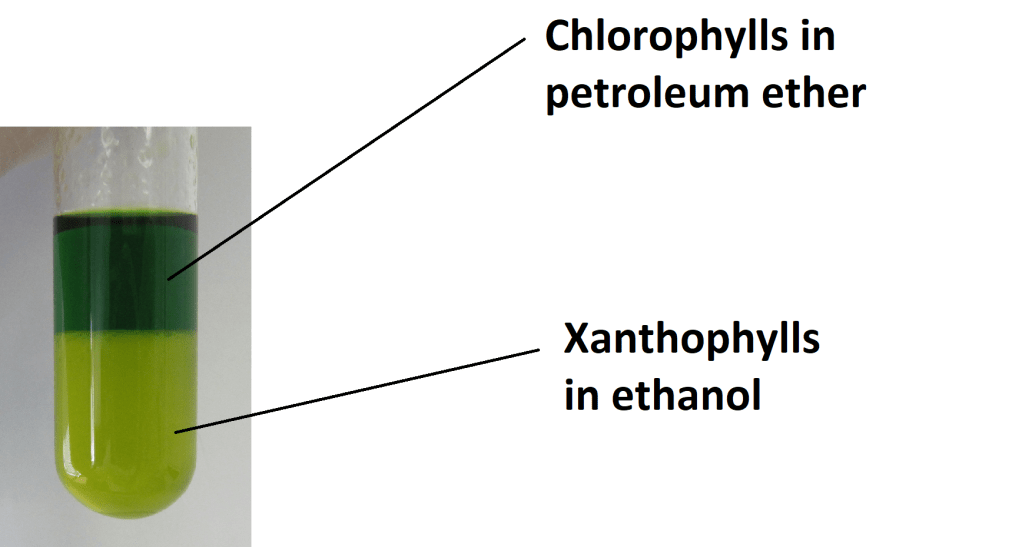
The property dictating into which solvent the individual components will go, is solubility of the compounds in the different solvents. Also the polarity must be considered to carry out the extraction successfully. The polarity of the solvent and the compound being extracted must be similar for the extraction to occur. In the case of natural dyes, apolar solvents (e.g. ethanol, petroleum ether) are used since Chlorophylls, Xanthophylls and Anthocyanins have apolar character.
The experiment
Aim: to detect what natural dyes are present in leaves samples
You will need:
- ethanol
- petroleum ether
- cotton wool
- mortar with pestle
- filter sand
- 10x filtration paper
- 5x test tubes
- 5x Petri dish
- glass rod
- filtration apparatus (stand, beaker, filter funnel, iron ring)
- plastic pipettes
- 5x different types of leaves (green, yellow, red)
Procedure
Firstly, set up the filtration apparatus – attach an iron ring to the stand, put a funnel into the ring and a beaker below (see image 1). Fold a filter paper and put it in the funnel.
Fold a filter paper in half and cut a small hole in the middle. Make a small wick from cotton wool and insert it through the hole (see image 2).
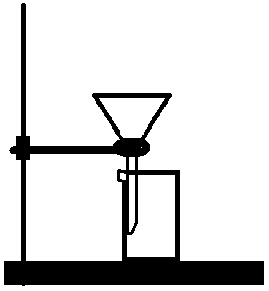

Tear apart your leaves (only one type at a time) to as small pieces as you can and put them in the mortar. Add a bit of filter sand so the leaves can be smashed better and 15ml of ethanol in aliquots (use plastic pipette). Once thoroughly smashed, pour the solution into the funnel with filter paper using a glass rod so you don’t spill anything and wait for it to filter.
Pipette 3ml of the filtered solution into a test tube and the rest into a petri dish.
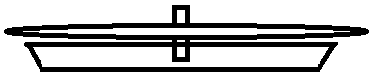
Take your pre-prepared filter paper with cotton wool and carefully put it into the petri dish so only the bottom of the wick touches the solution (see image 3). Make sure the filter paper is NOT in the solution. The solution will be slowly drained up through the wick and form a chromatogram. After the chromatogram is finished, carefully remove the filter paper from the wick and let it dry.
Next, add 2ml of petroleum ether into the test tube with your solution. Carefully shake it and observe the separation of individual dyes into the two solvent layers.
Repeat the same procedure for all your leaves and fill in the required section of the worksheet. If you are using the same filter funnel or any other glassware, wash it before you start another measurement so you obtain as accurate results as possible.
Results
The outcome depends on whether you run the experiment in spring or autumn.
Spring
We tested 5 different leaves:
| Sample NO. | Name | Chlorophylls | Anthocyanins | Xanthophylls & Melanins |
| 1 | Berberis thunbergii | ✓ | ✓ | X |
| 2 | Acer japonicum | ✓ | ✓ | ✓ |
| 3 | Apple tree (Malus domestica) | ✓ | X | ✓ |
| 4 | Prunus avium | ✓ | X | ✓ |
| 5 | Prunus domestica | ✓ | X | ✓ |
All leaves had chlorophyll present. Berberis thunbergi and Acer japonicum were the only ones that had Anthocyanins.
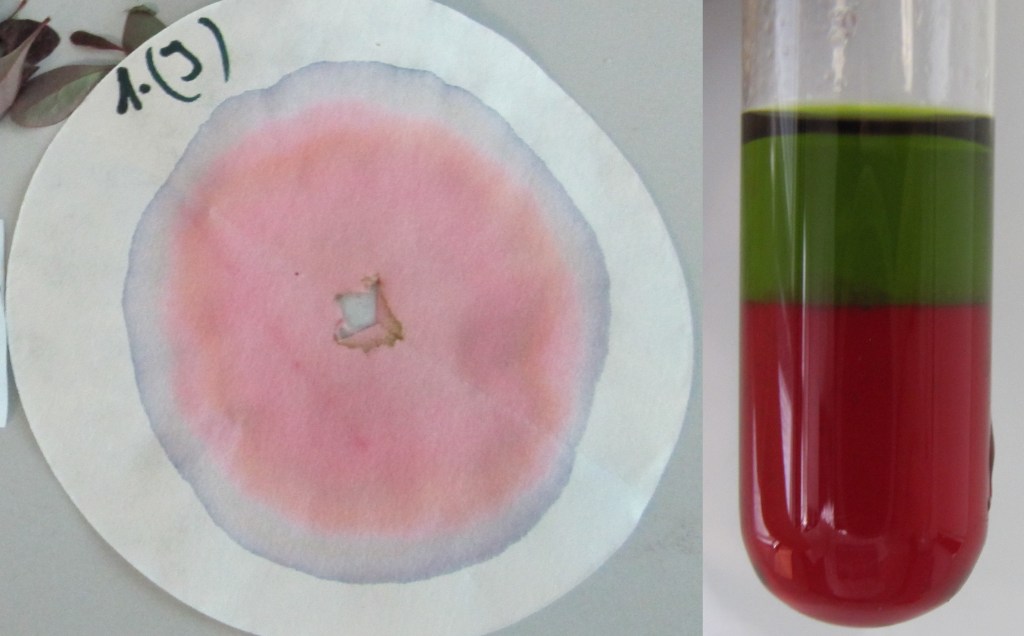

Autumn
Again, we tested 5 leaves – the same ones we did in spring.
| Sample NO. | Name | Chlorophylls | Anthocyanins | Xanthophylls & Melanins |
| 1 | Berberis thunbergii | ✓ | ✓ | X |
| 2 | Acer japonicum | ✓ | X | ✓ |
| 3 | Apple tree (Malus domestica) | ✓ | X | ✓ |
| 4 | Prunus avium | ✓ | X | ✓ |
| 5 | Prunus domestica | ✓ | X | ✓ |
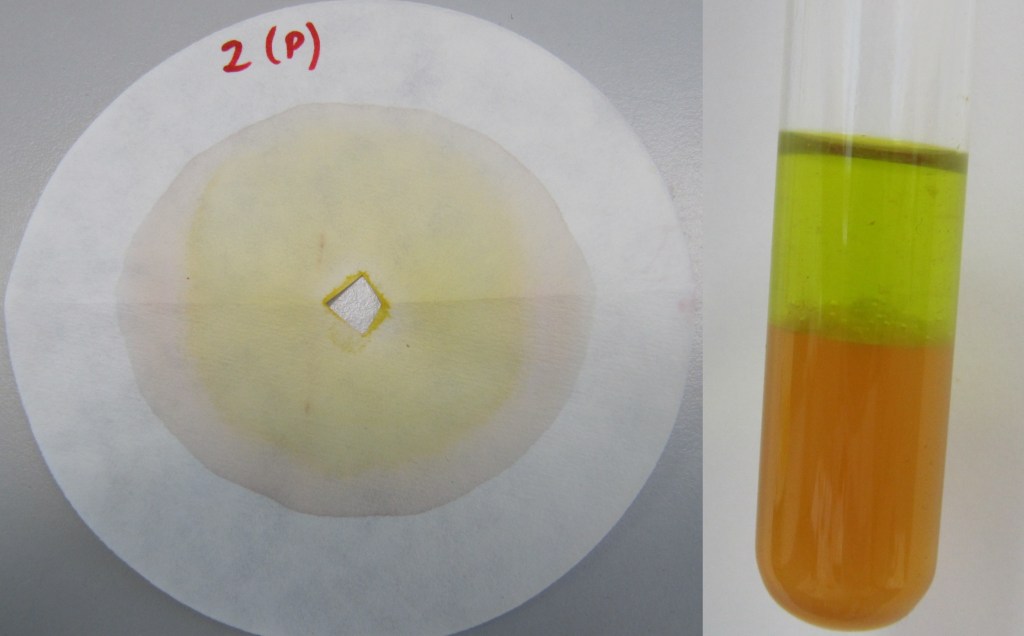
All leaves had chlorophyll present and melanins (mixed with xanthophylls) were also mostly present. As you can see in the picture below, the chlorophylls present have yellow-ish colour. This is due to the fact that in autumn chlorophylls begin to degrade slowly in plants.
In the research lab

This work is licensed under a Creative Commons Attribution 4.0 International License.