Download a printable version of this document here
Enantiomers are defined as a pair of stereoisomers that are non-superimposable mirror images of one another. This means they are molecules that are made up of identical atoms, bonded together in the same way, i.e. they have the same connectivity. And yet, the 3D arrangement of the atoms in enantiomers are different, as these molecules are mirror images of each other. You cannot superimpose one onto the other without breaking and remaking bonds. A pair of enantiomers can be seen below. Note the central atom is described as the stereogenic centre (or stereo-centre).

If a molecule has the property of having a non-super imposable mirror image it can be described as being chiral, i.e. possess chirality. Structure and shape often determine the way in which a molecule interacts with other molecules, and we see this in nature all the time. Many of the receptors within our body that respond to drugs are chiral, or isomeric, and therefore the chirality of the drug is incredibly important. A simple example of this and more is in the chemBAM experiment seen here.

Diastereomers are defined as stereoisomers with more than 1 stereo-centre that are non-superimposable non mirror images of one another. Again, this means they are molecules that are made up of identical atoms, bonded together in the same way, i.e. they have the same connectivity. But the 3D arrangement of the atoms in diastereomer are different, as these molecules are non-mirror images of each other and contain more than one stereocentre. You also cannot superimpose one onto the other without breaking and remaking bonds.
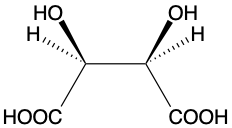
Some molecules can be described as achiral. This is true when a would-be chiral molecule also possesses a plane of symmetry (see Mesotartaric acid left). These molecules are also described as meso-compounds. Achiral is not the same as non-chiral. Non-chiral molecules possess no stereocentre and are not optical isomers.
Page Author; Thomas Squire

This work is licensed under a Creative Commons Attribution 4.0 International License.