
Download a printable version of this document here
Enzymes are very efficient and highly selective natural catalysts. So is it possible to design synthetic enzymes which can be designed to catalyse a specific reaction in aqueous conditions? This is the question scientists such as those at the University of Birmingham are trying to solve.
Enzymes by their very nature have complex structures on the molecular level. Thus it is extremely difficult to replicate this complexity under laboratory conditions. Thankfully simpler amino acid structures such as coiled coils (see below) can be synthesised in a lab which can act as artificial enzyme scaffolds. These coiled coil structures are made up of naturally occurring amino acids, which can be designed to fold into the coiled coil by taking into account the hydrophilic/hydrophobic nature of the different amino acids. Hydrophobic amino acids come together at the centre of the coiled coil to form a hydrophobic pocket, whereas the hydrophilic amino acids are on the outside of the coiled coil interacting with the aqueous solvent.
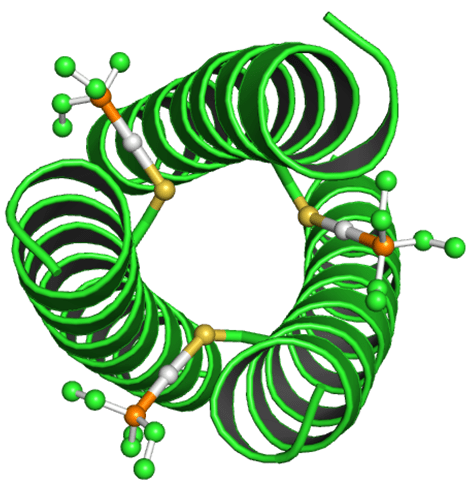
Many enzymes contain transition metal centres which are often the source of an enzymes catalytic activity. However “nature” only uses a small number of transition metals, mostly limited to 1st row transmission metals. Synthesising artificial enzymes allow a wider variety of transition metals to be utilized, with the current focus on 2nd and 3rd row transition metals such as gold, silver and platinum with known catalytic activity.
Incorporating transition metals, especially larger 2nd and 3rd row transition metals, into the artificial enzyme scaffold (coiled coils) can be difficult. This is because incorporating the metals into the hydrophobic pocket can upset the delicate balance of hydrophilic/hydrophobic constituents. It is however possible to use artificial/un-natural amino acids which can bind to the metal and also add some stability to the artificial enzyme scaffold (coiled coil).

Natural enzymes also have a pocket to selectively bind a desired substrate. This pocket is usually highly specific and can contain numerous amino acids correctly positioned to ensure the binding is selective. This is another challenge the relatively small artificial enzyme template (coiled coil) must overcome to function as a true artificial enzyme.
There are also some drawbacks associated with natural enzymes such as the fact that they are only stable in a narrow temperature range and they do not tolerate changes in their environment. Again the artificial enzyme scaffold (coiled coil) can be modified with artificial/un-natural amino acids to counteract these traditional enzyme problems though improving the artificial enzyme scaffold (coiled coils) stability.
The aim of designing successful artificial enzymes is to pave the way for next generation of catalysts which akin to natural enzymes have high selectivity/efficiency under mild reaction conditions, including aqueous conditions. This type of catalyst could have numerous applications from improving the production/synthesis of drug like molecules, to improving the efficiency of industrial scale synthesise.
To learn more; see this article written in the conversation about artificial proteins based upon research undertaken by Dr Anna Peacock and her group.
Go back and learn more about the chemistry of catalysis here.

This work is licensed under a Creative Commons Attribution 4.0 International License.