Download a printable version of this document here
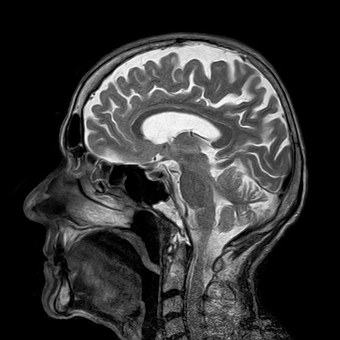
Chemists use the phenomenon of ‘magnetic resonance’ all the time, and these are the ‘MR’ in MRI, and also is ‘NMR’. When an atom is placed within a magnetic field, if that atom has an odd number of protons, or an odd number of neutrons, such as hydrogen, it can ‘resonate’ between different energy states, if supplied with enough energy from a radio wave.
A water molecule has two hydrogen atoms that are covalently bonded to oxygen – both hydrogens are chemically equivalent, and behave in the same manner. The hydrogen nucleus (often called a proton, because it is just a proton), can exist in two energy states, either spin up or spin down. In the absence of an external magnetic field, it doesn’t matter which state the proton is in, as they have the same energy. However, once we put the proton into a magnetic field, it can be either aligned with the field (which is easier), or aligned against the field (which is more difficult). These two states are no longer equivalent, as it takes more energy to be aligned the wrong way around.
Our body is made up of 60 % (by weight) water, so we have a lot of these little bar magnets in our body. When you lie in an MRI tube, you are lying inside a very strong magnet, and your water protons can be either aligned or aligned anti-parallel with the field. Whilst you are in the MRI tube, your body is subject to radio waves, which as you know penetrate buildings, and go right through us; if they did not, we wouldn’t receive much of our communication. A short, intense pulse of radio energy is applied across the whole body in the MRI scanner, which is absorbed by all of the protons in the body. After this pulse, the protons emit radio waves, which can be detected, just like NMR.
In NMR – we put our sample in an NMR tube, and place it in magnet; this is exactly the same as the MRI magnet, but it is usually the other way around – we don’t tend to want to make patients stand or ‘hang’ vertically! We use NMR to tell us information about the molecules in the sample tube. If we are looking for protons, then we excite the sample with radio waves, to make all the protons resonate; and then we collect the radio signal from all the protons until they stop resonating. Different protons will be in different chemical environments, so they will resonate at slightly different frequencies. In NMR we use the different frequencies of radio waves given off by the sample to determine the chemical structure, and we get a spectrum.What would the NMR spectrum of a body look like? Well mostly we would see only water; there are lots of lots of protons within the body, in different chemical environments… proteins, DNA, fat, sugar… but most of these are in very tiny quantities, compared with water. Knowing that the patient contains water, is not overly useful as a diagnostic technique!
MRI is different to NMR – in that we are using the technique to build up an image; we are mapping water. Because the spectrum is rather simple (predominantly one signal from the water protons) we can apply a magnetic gradient across the patient in the tube, so that they are not in an even magnetic field. When this is done, the frequency at which a proton resonates when excited with radio waves, now depends on where it is.
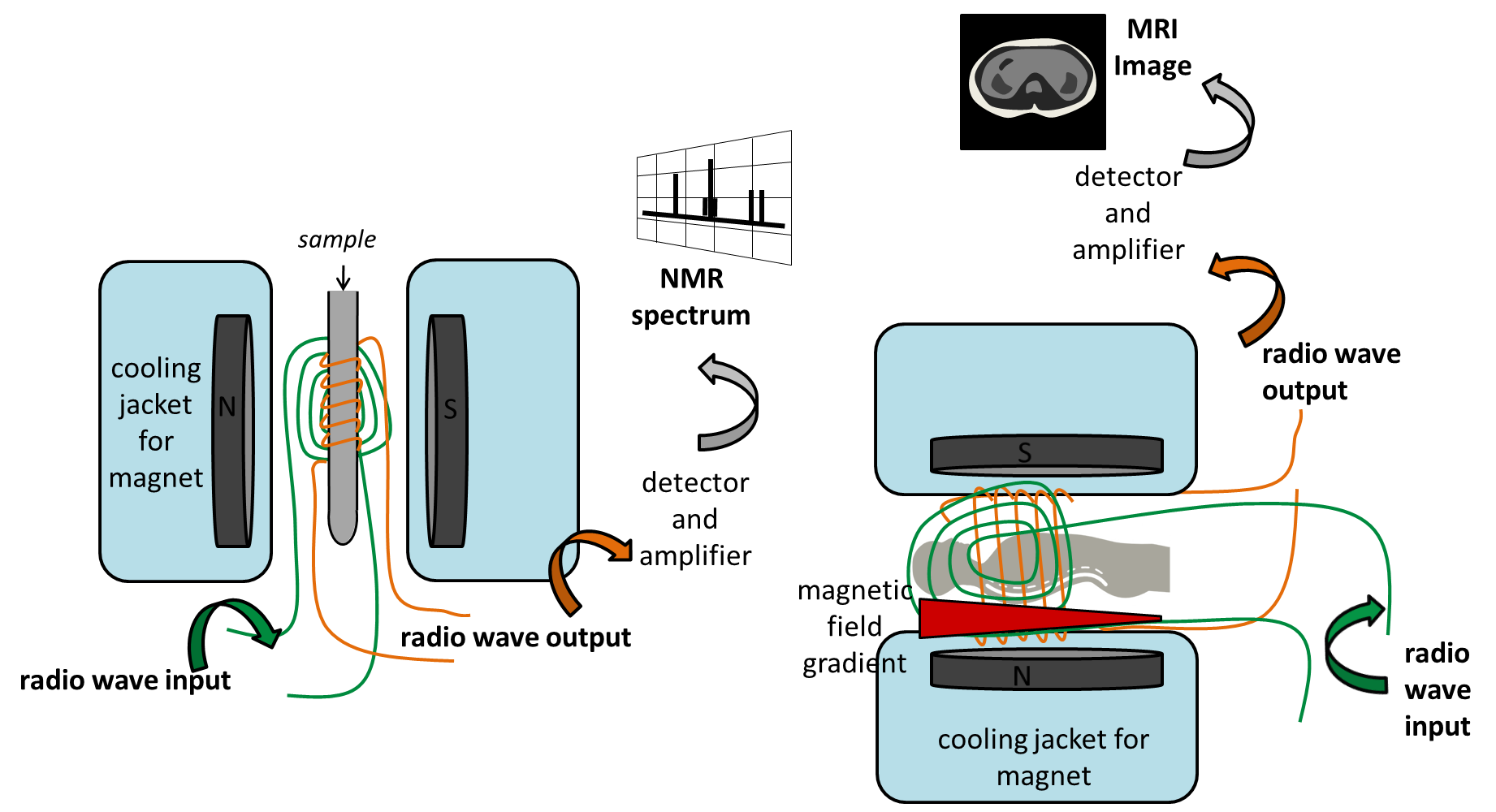
Protons at the feet are in a lower magnetic field, for example, than those at the head, and therefore they resonate at a lower frequency. The protons at the head are in a higher magnetic field, and resonate at a higher frequency. Therefore we can tell that we have water at different places in the body, and we can use this to build a picture. The tissue that has more water will appear brighter, and you start to build up an image of the water in the body. If you want to read more about the basics of MRI, take a look at the following article written in the conversation.
In the clinic, x-rays offer us the ability to image the anatomy quickly, and relatively inexpensively, but there are several reasons why an MRI may be preferred. Firstly, MRI does not use ionising radiation, like x-rays, and is often thought to be a safer imaging option. That said, the radiation dosage from a single x-ray scan is very low. X-rays give contrast in images by being reflected off bone and dense materials more than soft tissue, and hence are great for visualising bones, but it is more difficult to determine information about tissue and organs. MRI is an excellent imaging technique that allows us to build up 3D images of the body, with the ability to image deep within the patients tissue, safely and without surgery. We can see inside patients in great detail without opening them up.
Back to top of page
Contrast in MRI, and the role of the chemistry researcher
An MRI scan of a body will reveal a lot of anatomical information, much more than an x-ray, because different tissues contain different amounts of water, and will appear as brighter pixels where there is more water, and darker pixels where there is less water. The contrast of the image can be made sensitive to different things though, not just the concentration of the water, by changing the lengths of time that the radio waves are collected, and other scan parameters. This can be very useful for highlighting different parts of the body.

Many chemistry researchers work in the area of developing MRI contrast agents. These are chemicals that are injected into patients that can be seen in an MRI scan, in order to improve the visibility of internal body structures. The most commonly encountered compounds that are used in the clinic contain gadolinium(III) (Gd3+) complexes. This is because Gd3+ contains 7 unpaired electrons, with the electronic configuration [Xe]4f7, each electron is in a different f-orbital. Just like unpaired protons and neutrons, unpaired electrons also possess ‘spin’, and Gd3+ have 7 electrons, each contributing to the spin of the metal centre, so it behaves like quite a strong magnet for a single ion. When water protons in the body are in close proximity to gadolinium contrast agents, they are affected by the movement of these strong magnet centres, and relax back to their ground state more quickly, after being excited with radio waves. MRI imaging systems can be set up to show bright spots where the protons relax back to the ground state quickly, and the contrast agent can then be seen in the image. To read more about chemistry research into new contrast agents for MRI from the group of Dr Anna Peacock, take a look at the following article written in the conversation.
MRI is not only used for imaging people for medical applications. Chemistry researchers at the University of Birmingham, working in the group of Dr Melanie Britton, use MRI techniques to understand molecular processes that underpin systems found in a range of applications from structured materials, manufacturing and energy storage, to medical applications relevant in the diagnosis of cancer and the development of biomarkers. Currently, they are interested in the visualisation of electrochemical processes in batteries, corrosion and electroplating; the visualisation of chemistry in flow and heterogeneous media; probing colloidal stability; the development of MRI contrast agents and the characterisation of reverse micelles.

Mapping corrosion by MRI. Image taken from published research which can be accessed here.
Back to top of page

This work is licensed under a Creative Commons Attribution 4.0 International License.