
Download a printable version of this document here
Atomic orbitals can seem like a daunting prospect for someone new to the idea, however it is useful to know about as it can help with other aspects of chemistry. This is because the shapes of the orbitals and how they interact with one another underpins a lot of chemistry, particularly the bonding between molecules and how molecules react with each other.
Orbital shapes and sizes
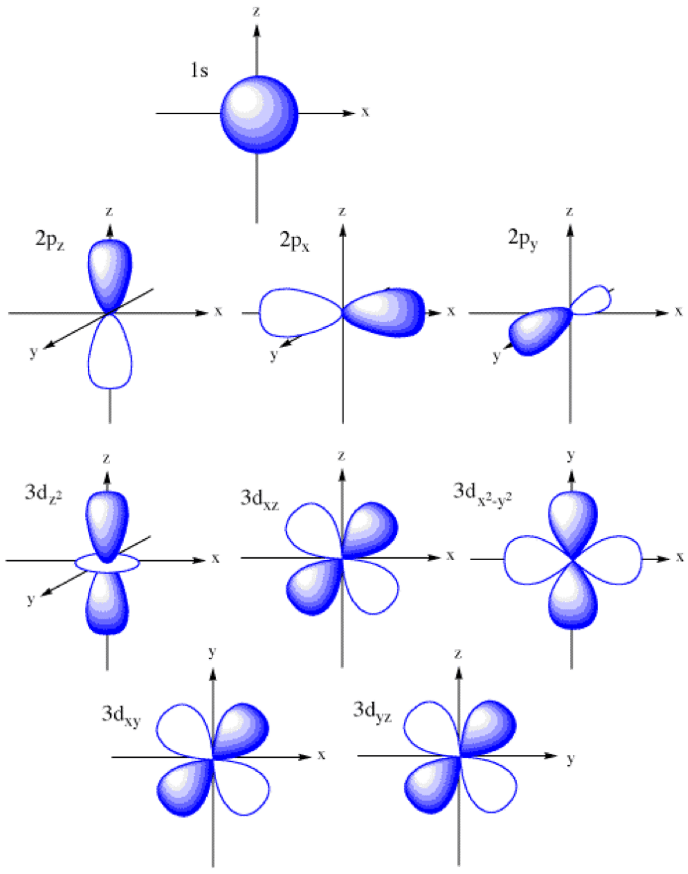
To start with only atomic orbitals will be discussed. Atomic orbitals are the place in an atom where the electrons dwell. They are often described in terms of shells and subshells in alevel. A shell more broadly speaking describes the energy of an orbital (1,2,3,4,5 etc) and a subshell describes the shape of an orbital (s,p,d). As the energy of an orbital increases (i.e. the shell number) from 1 to 2 to 3 etc, the size of the orbital increases. NOT the shape. The shapes of the 3 orbitals can be seen below;
Filling orbitals
There is a specific order in which orbitals must be filled which may seem daunting at first. However, this is where a periodic table becomes very useful. See the video which shows how using a periodic table can help in organising electrons in the correct orbital – also termed the electronic configuration. It’s important to remember only 2 electrons can occupy 1 orbital at a time.
Once you combine the knowledge of the orbital sizes/shapes and how to fill them, a lot of chemistry becomes easier to explain!
Hybrid orbitals
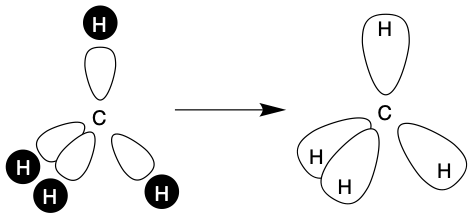
Again hybrid orbitals sounds like a complicated idea, however hybrid orbitals are only atomic orbitals combined to together. Here we are going to discuss sp, sp2 and sp3 orbitals seen below. As the names suggest these hybrid orbitals are made up of 1 s orbital and either 1, 2 or 3 p orbitals. Hybrid orbitals are an important concept when trying to understand the shapes of molecules and how the atoms in a molecule interact/bond.

These hybrid orbitals directly link to how many valence electrons are present in the atom and the atoms surround/bond to the atom (Note valence electrons are electrons in an atoms outer shell). For example the molecule CH4 and hydrogens surrounding the central carbon which has 4 valence electrons. Therefore 4 orbitals are needed to form the 4 bonds using the 4 electrons from the carbon. Thus, the carbon needs four sp3 orbitals so combines its one s orbital with its three p orbitals to get the four sp3 hybrid orbitals.
An example of using this concept is with Lewis acids/bases, especially how BF3 reacts with NH3. In BF3 the central B atom has 3 valence electrons so forms 3 sp2 orbitals. There is however 1 p orbital that is not used. This free p orbital is free to accept the pair of electrons from NH3, therefore allowing BF3 to act as a Lewis acid.

Bonding orbitals
Bonding orbitals combine orbitals of different atoms and are the basis of covalent bonds. There are 2 types of bonding orbitals shown below. The sigma bonding orbital; which makes up single covalent bonds. And pi bonding orbitals; which make up double bonds (also described as pi bonds). The important concept here is that the orbitals of the two atoms must align to allow sufficient overlap in 3D. The overlap of the two orbitals is what forms “a bond”.

For every bonding orbital there is an antibonding orbital. These antibonding orbitals are always the opposite to an antibonding orbital and signified with a *. Antibonding orbitals are important in describing how a reaction takes place through breaking then making new bonds. The antibonding orbital effectively acts as a target for a pair of electrons to attack. When the new pair of electrons interact with the antibonding orbital, it forms a new bond and forces the corresponding bonding orbital (or corresponding bond) to break apart. A good example is the nucleophilic substitution reaction (or SN2 reaction) as seen below.

To learn more generally about bonding and intermolecular forces, see the chemBAM page.

This work is licensed under a Creative Commons Attribution 4.0 International License.