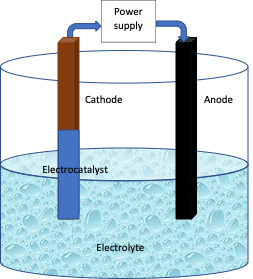
Electrocatalysts are the materials used to increase the rate of electrochemical reactions. There are many electrochemical reactions that are theoretically possible but need more energy than predicted to be accomplished practically. Examples of a few such electrochemical reactions are water splitting, CO2 reduction, ethanol oxidation, and methanol oxidation. Electrocatalysts are very useful in these cases to decrease the energy barrier. Electrocatalysts can also be used for the synthesis of organic molecules and polymers. The efficiency of the electrocatalysts can be tuned by modifying the structure, composition, and surface of the material.
There are two types of electrocatalysts, heterogeneous and homogeneous electrocatalysts.
Heterogeneous electrocatalysts are catalysts that are in a different phase than the reactants. They function at the surface of the electrode or the electrode itself acts as the catalyst. In the former case, the catalyst material is coated on the electrode either anode or cathode, or both as needed, and the coated electrodes are inserted in the cell. Metals, metal oxides, metal carbides, etc are used as heterogeneous electrocatalysts. The Chloralkali process is an example of a large-scale electrochemical reaction that uses a dimensionally stable anode coated with an electrocatalytic layer of mixed titanium and ruthenium oxide to generate NaOH and chlorine.
Homogeneous electrocatalysts are catalysts that are in the same phase as reactants or soluble in the reaction medium. Enzymes and coordination complexes are examples of homogeneous electrocatalysts.