Background
Colorectal cancer is the third most common cancer in men and women worldwide. Almost 55% of cases occur in the more developed regions of the world, and recent scientific research suggests that a contributing factor could be excess iron in the intestine from a diet rich in red meat.
Iron is essential for our diet. For example, haemoglobin in red blood cells contains iron and is crucial for oxygen transport around the body. However, too much iron in the large intestine is believed to cause the formation of tumours.
Chemists and cancer scientists working with Chris Tselepis at the University of Birmingham are looking at ways to stop iron causing damage in the large intestine. The aim is to develop a drug that will bind iron in the large intestine to prevent the onset of cancer. This is called a ‘prophylactic‘ treatment.
In order to achieve this, researchers are currently looking at a biological polymer (a biopolymer), called alginate. This biopolymer binds iron, and also meets some of the requirements needed for a drug to be active in the large intestine.
Alginates
 Alginates are an example of a structural biopolymer. Structural biopolymers are polymers that are produced by living organisms. They provide plants with their strong stems, crustaceans with their hard shells and our bodies with flexibility and movement.
Alginates are an example of a structural biopolymer. Structural biopolymers are polymers that are produced by living organisms. They provide plants with their strong stems, crustaceans with their hard shells and our bodies with flexibility and movement.
Biopolymers are extracted for use in the food industry. Gelatin is the most familiar – it is used in sweets and puddings for its ability to form gels. Alginates are extracted from seaweed and used as thickeners and stabilisers (E400-E404).
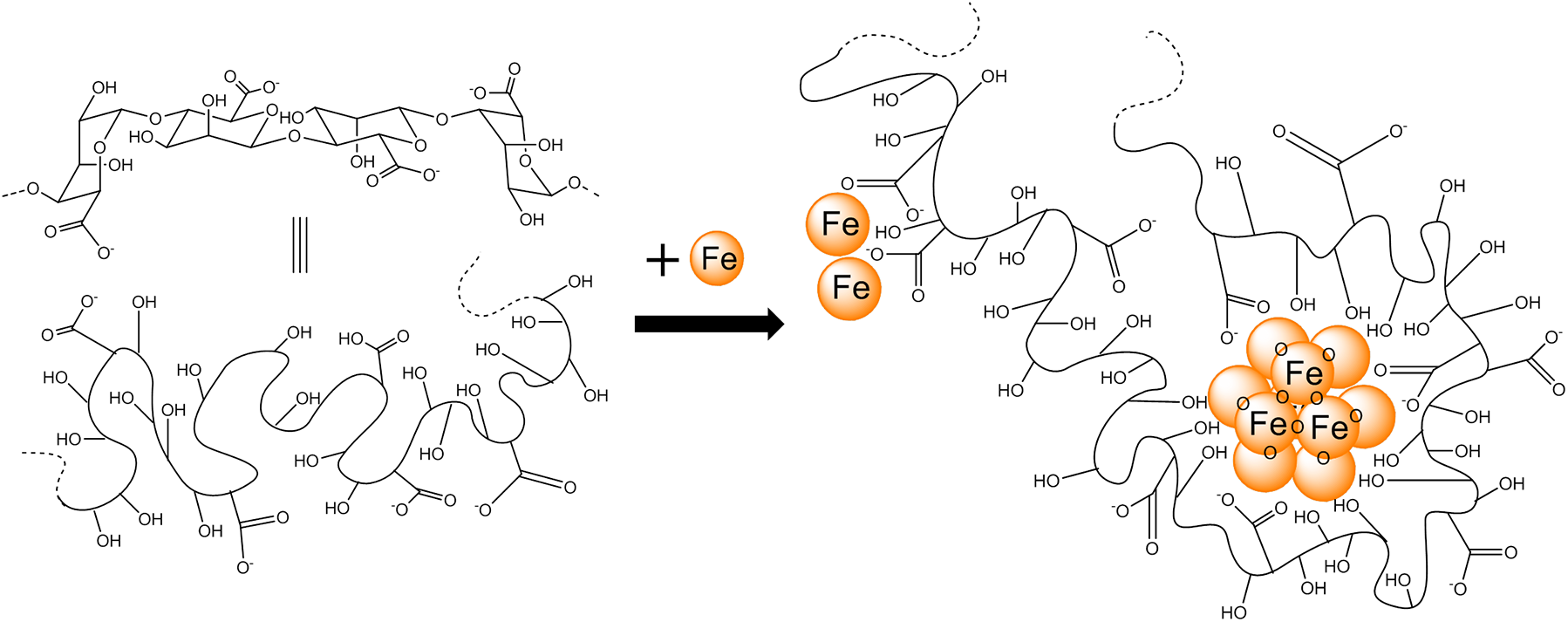
Alginates are polymers made from several hundred sugar (saccharide) monomers. Alginates are copolymers. They contain two types of monomer, known as M (β-D-mannuronate) and G (α-L-guluronate). The polymer is made up of blocks of identical or alternating monomers  (MMMMMM, GGGGGG or MGMGMG). Different species of seaweed produce alginates with different composition.
(MMMMMM, GGGGGG or MGMGMG). Different species of seaweed produce alginates with different composition.
Alginates have a carboxylic acid functional group on each monomer. These are easily deprotonated (lose H+) to form carboxylate groups. These negatively-charged carboxylate groups bind to positively charged metal ions. In the sea, alginate takes up calcium ions (Ca2+) from the seawater.
Where there are sections of the alginate polymer with lots of ‘G’ segments, they bind calcium ions particularly strongly. The calcium ions are believed to line up between GGGGGG sections, a bit like eggs in an egg box.
If alginate is to be used to bind iron in the gut, it needs to be able to bind to iron (Fe3+) ions. Ideally, we want an alginate that will bind to iron but leave the calcium (an important nutrient) alone. Researchers at the University of Birmingham have been investigating alginates with lots of ‘M‘ units for this purpose.
Exercise 1: identifying common organic functional groups in polysaccharide biopolymers.
Exercise 2: alginate origami.
(See activity resources at top of page for downloads)
The experiment
AIM
To determine whether a high-M or high-G alginate is better for selectively absorbing Fe3+ ions.
YOU WILL NEED
- 6 small beakers

- plastic pipettes
- solution ‘High G’ alginate (Manugel GHB) (aq) (plus food colouring)
- solution of ‘High M’ alginate (ProtaSea AFH, or Manucol LD ) (aq) (plus food colouring)
- 0.1% w/v CaCl2 (aq) solution
- 0.1% w/v FeCl3 (aq) solution
PROCEDURE
Comparing the binding of calcium ions:  Pour 25 mL of CaCl2 solution into two beakers, and label them G and M. Using a plastic pipette, suck up 1-2 mL of the viscous ‘High G alginate’ solution, and pipette it dropwise into the CaCl2 solution, in the beaker marked G. Repeat this with the ‘High M alginate’, in beaker M, and observe the difference – swirl the beakers for a few seconds. You can tip the gels into your hand, over a sink, to see the differences.
Pour 25 mL of CaCl2 solution into two beakers, and label them G and M. Using a plastic pipette, suck up 1-2 mL of the viscous ‘High G alginate’ solution, and pipette it dropwise into the CaCl2 solution, in the beaker marked G. Repeat this with the ‘High M alginate’, in beaker M, and observe the difference – swirl the beakers for a few seconds. You can tip the gels into your hand, over a sink, to see the differences.
 Comparing the binding of iron ions: Pour 25 mL of FeCl3 solution into two beakers, and label them G and M. Do not get this solution on your hands as it is acidic and corrosive. Using a plastic pipette, suck up 1-2 mL of the viscous ‘High G alginate’ solution, and pipette it dropwise into the FeCl3 solution, in the beaker marked G. Repeat this with the ‘High M alginate’, in beaker M. Gently swirl the solutions for a few seconds -what happens in each case?
Comparing the binding of iron ions: Pour 25 mL of FeCl3 solution into two beakers, and label them G and M. Do not get this solution on your hands as it is acidic and corrosive. Using a plastic pipette, suck up 1-2 mL of the viscous ‘High G alginate’ solution, and pipette it dropwise into the FeCl3 solution, in the beaker marked G. Repeat this with the ‘High M alginate’, in beaker M. Gently swirl the solutions for a few seconds -what happens in each case?
Control experiment: Pour 25 mL of deionised water into two beakers, and label them G and M. Using a plastic pipette, suck up 1-2 mL of the viscous ‘High G alginate’ solution, and pipette it dropwise into the beaker marked G. Repeat this with the ‘High M alginate’, in beaker M. Gently swirl the solutions for a few seconds – what do these two solutions look like?
If you wish to conduct this experiment just to see the gelling of the ‘High G’-type alginates, it can be adapted using Sodium Alginate purchased from a chemical supplier, such as Sigma Aldrich, or a specialist food supplier e.g. http://www.souschef.co.uk/sodium-alginate.html. Make the alginate solution up, adding solid to stirring water, until a viscous, but pourable consistency is reached. (The amount of alginate needed to be added will vary, in the range 0.5-3 g per 100 mL of water). More information can be found here.
The actual alginates needed to run the experiment are not commercially available but if you contact us, we can send you some (within the UK).
QUESTIONS
- What do the two alginates look like when they interact with calcium ions? Can you pick up the gel balls?
- Which alginate binds more strongly with calcium ions?
- What do the two alginates look like when they interact with iron ions?
- Look at the control solutions, where you have just mixed the alginates with water – what happens? What does this confirm?
- Which of the two alginates do you think will act best as a drug to bind iron, in a calcium-rich environment?

From left to right: High G alginate in water, Fe(III) solution, Ca(II) solution, blank Fe(III) solution, blank Ca(II) solution, High M alginate in water, Fe(III) solution, Ca(II) solution.
In the research lab

In the School of Cancer Sciences at the University of Birmingham, researchers, including Dr Richard Horniblow, working in the group of Dr Chris Tselepis, are currently testing a ‘high M‘ alginates in clinical trials. Read about this work in the Daily Mail article, and find out more about the group’s research here.
Publications relating to this work

To access this paper click here.

Abstract above from published article at: http://onlinelibrary.wiley.com/doi/10.1002/mnfr.201500882/abstract
Page author: Nicola Rogers Simpson
Back to top of page
This work is licensed under a Creative Commons Attribution 4.0 International License.