
Download a printable version of this document here
Covalent bonding
A covalent bond is a bond which occurs when 2 atoms share a pair of electrons. It is symbolised using a straight line between 2 atoms. Double and triple covalent bonds in akenes and alkynes for example are formed of 2 or 3 covalent bonds (i.e. whereby 2 or 3 pairs of electrons are shared).
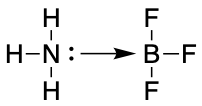
Another type of covalent bond is the coordinate (covalent) bond also known as a dative bond. Here the pair of electrons is donated from one atom to another atom which is electron deficient. This is analogous to the Lewis bonding model (see chemBAM page here to learn more). It is usually symbolised when appropriate using a straight arrow coming from the electron rich atom towards the electron deficient atom.
Covalent bonds make up the backbone of all molecular structures from diatomic molecules such as O2 to macromolecular structures such as diamond and buckminsterfullerene (bucky-balls). To go deeper into the theory behind bonding with atomic and molecular orbitals go to the chemBAM page here.
-
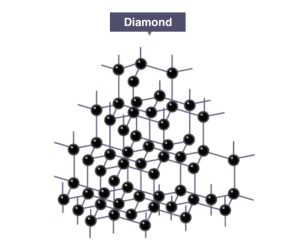
-

Bucky-ball
image source (left).
Ionic bonding

Ionic bonding is an electrostatic attraction which forms between 2 oppositely charged ions. This predominantly occurs in the form of a lattice which is formed of many oppositely charged ions in the form of a crystal structure. An example of this is common salt NaCl which has a lattice made up of Na+ cations and Cl– anions.
Metallic bonding
Metallic bonding occurs through electrostatic interactions between a lattice of positive ions (cations) and a delocalised “sea of electrons”. It is the “sea of delocalised electrons” which allow some metals to conduct electricity as the electrons are free to move within the metal. Therefore a good question is why don’t all metals conduct electricity? The answer is complex (and is only fully taught at graduate level), but involves the individual properties of metallic elements such as; how many valence (or free) electrons there are per atom and how much “space” there is for the electrons to move freely. These are the main factors in the quantum mechanical explanation known as band theory.
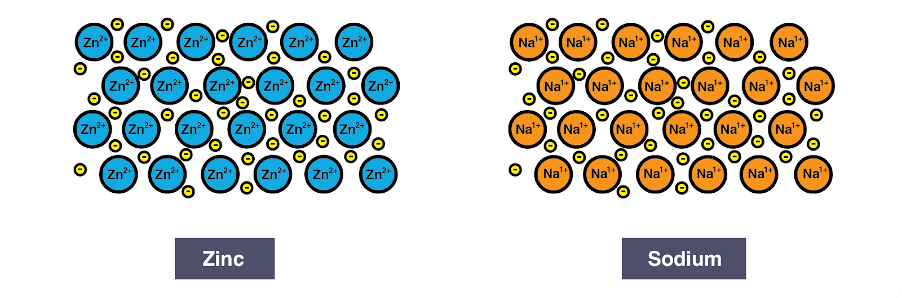
Image source.
Bonding vs intermolecular forces vs intramolecular forces
The precise difference between bonding and intermolecular forces is quite vague. This is why many explanations usually take covalent bonds vs intermolecular forces, as covalent bonds rely on sharing of a pair of electrons to form a “physical” bond. Whereas intermolecular forces rely on a “force” to bring atoms or molecules together. Thus in this type of explanation ionic bonding can often be described as being an intermolecular force, as it is the electrostatic interaction between the ions that holds the lattice together. However technically covalent, ionic and metallic bonds are all formed through intramolecular interactions (i.e. interactions between individual atoms) and therefore are described as intramolecular forces. Intermolecular forces technically refer to forces between molecules. These intermolecular forces are discussed next and are all weaker than intramolecular forces.
Permanent dipole–dipole forces
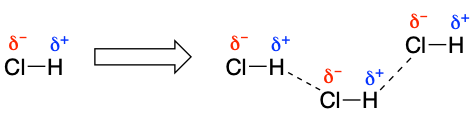
A permanent dipole-dipole intermolecular interaction relies on a molecule having a permanent dipole. But what is a permanent dipole? A permanent dipole occurs within a molecule when it has atoms with large differences in electronegativity and thus its atoms have partial charges. For example H-Cl (see below), whereby the Cl is very electronegative (δ⁻) and the H not so much (δ⁺) , so a permanent dipole exists in the molecule. Because of this the HCl molecules can interact through permanent dipole-dipole intermolecular forces where the particle charged atoms in the molecules interact.
Induced dipole
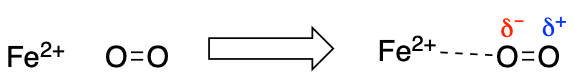
Some molecules can have an induced dipole if a highly electronegative atom is in close proximity. This then leads to induced dipoles interactions between the molecules. An example of this is a diatomic gas such as O2interacting with a charged ion such as Fe2+.
Dispersion (van der Waals, London) forces
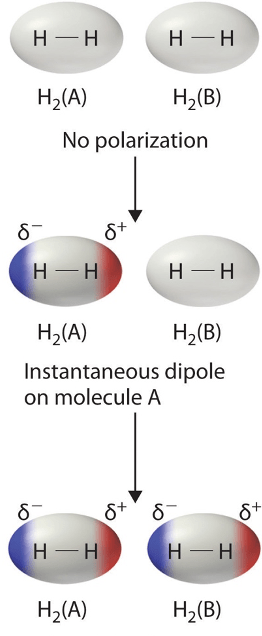
Dispersion forces also labelled van der Waals/London forces generally describe the interactions of molecules which contain atoms which are weakly electronegative. Even though these molecules contain atoms which are weakly electronegative, they can instantaneously produce an uneven distribution of electron density. This uneven distribution can cause interactions between molecules described as dispersion forces.
An example of this is a diatomic gas such as H2 interacting with other H2 molecules. Larger molecules have larger dispersion forces, however these dispersion forces are by far the weakest type of intermolecular interaction. Note these interactions are also sometimes described as induced dipoles in different curriculums due to one molecule inducing a dipole on another.
Hydrogen bonding
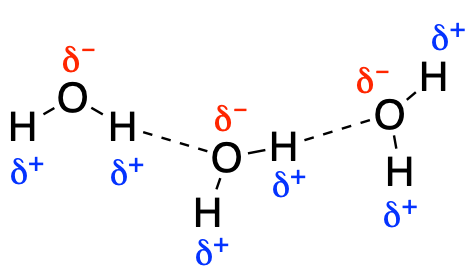
Hydrogen bonding is stronger than the other intermolecular forces discussed here, although it is not in fact a bond. It more resembles a specific/stronger version of the permanent dipole-dipole intermolecular interaction as it requires strongly electronegative atoms for it to take place. Hydrogen bonding only occurs between molecules which contain a polarised H atom (O-H ,N-H, F-H) and a another which contains a strongly electronegative atom (O, N, F).
Evidence of the effects of hydrogen bonding can be seen in the graph below. This highlights how the boiling points of the three molecules, (H2O, HF) which can undergo hydrogen bonding between themselves, are a lot higher than the trend would suggest (i.e. vs molecules that cannot undergo hydrogen bonding). Without the ability to hydrogen bonding these molecules would be predicted to have a lot lower boiling points.
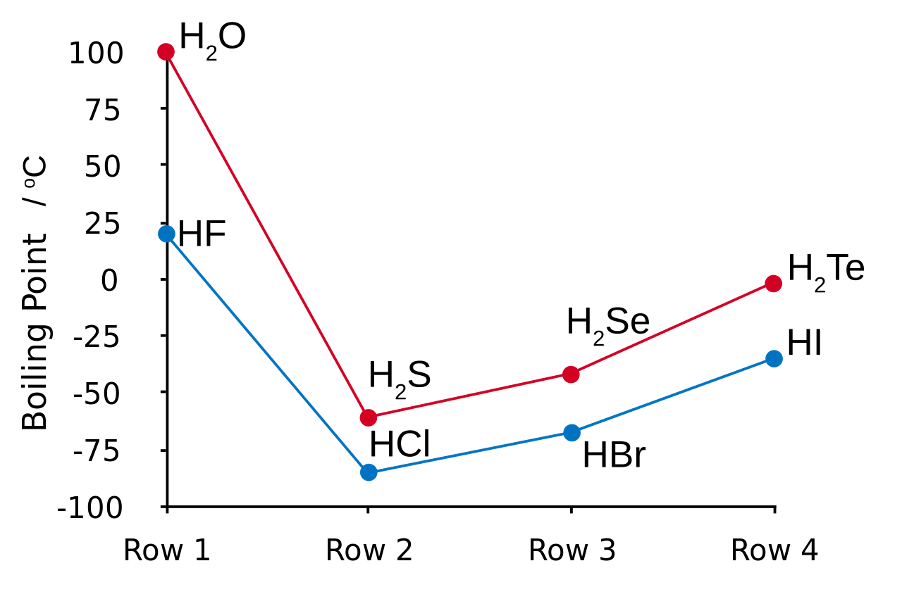
Note the reason for the general trend for the molecules that cannot undergo hydrogen bonding increases is, because the dispersion forces between molecules increase as the molecular weight of the molecule increases.

This work is licensed under a Creative Commons Attribution 4.0 International License.