
Download a printable version of this document here
IR (Infrared) absorption spectroscopy is a technique used by many researchers to both investigate and identify samples of interest. This is because the infrared spectrum of different functional groups within the molecules are unique so can be identified in a spectra. This can be seen in the spectra below of ethanol where the peaks corresponding to the CH and OH bonds can be seen. Also note the fingerprint region between 1500-500 cm-1 which is highly specific for different molecules, thus can be compared to known samples to identify sample tested. IR spectroscopy however is usually used alongside other techniques to confirm a substances identity. To learn more about identifying functional groups in a spectra see the link.

IR spectroscopy is also referred to as vibrational spectroscopy because this describes the underlying science behind how the spectra is produced. The IR radiation of a source is absorbed by a molecule, but molecules only absorb specific frequencies of IR radiation which allow the molecule to vibrate at the corresponding frequencies. By measuring which frequencies are absorbed/transmitted, an IR spectrum can be produced for the given molecule. Some of the ways a molecule can vibrate are seen below (with a molecule of the molecular formula CH2X2).



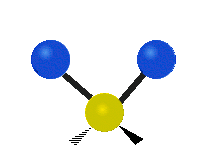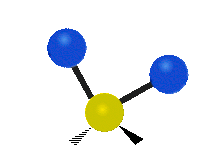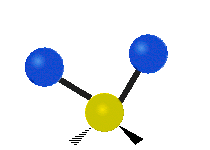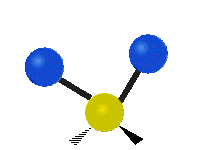
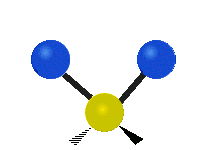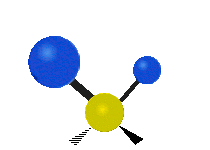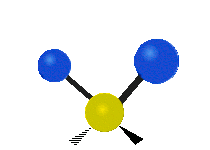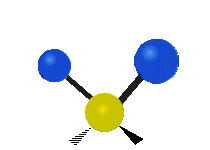
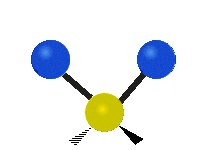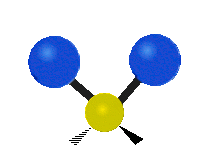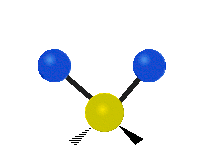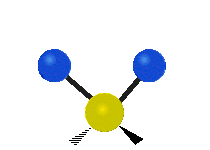

This work is licensed under a Creative Commons Attribution 4.0 International License.