Aims
To understand what a mixture is.
To learn that chromatography is a technique that scientists use to separate their mixtures.
To appreciate that the interaction between each component and the inside of the column determine how well the mixture separates and that components with a similar material to the inside of the column will stick to the column more strongly.
Background
When scientists perform reactions, they sometimes either have some of their starting material left in the mixture after the reaction or they have made some undesired product. This means that in order for them to obtain their desired product they must separate the mixture. This is done using a technique called Chromatography.
Chromatography can be done in a column:
One material sticks to the column better than the other material and the material that is good at sticking to the column is often a similar material to the inside of the column. This means only the material that doesn’t stick falls out of the column.
Task 1: Think about materials that stick together and materials that don’t stick together.
The tactile column
The tactile column has Velcro on the inside.

Task 2: Have a go mixing the plastic and woollen balls together and pouring them down the funnel and into the column. Feel which balls come out of the column. Which balls stick well to the column and which do not stick well?
Task 3: Suggest what else may stick to the inside of the column.
Extension (discussion about chromatography in a research lab):
Chromatography is a technique used to separate or purify mixtures.
Column chromatography involves a liquid mobile phase and a solid stationary phase.
The rate at which each component moves down the column is determined by its relative affinity for the mobile phase and stationary phase.

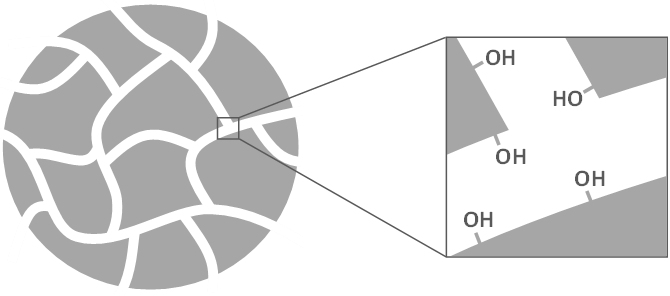
Silica:
Silica (SiO2) is a common stationary phase. You can find silica in sand on the beach. But the silica in columns is a bit different. It has lots of holes in it, called pores. The surface of all those holes is covered in Si-OH groups. We call these silanol groups. The holes give the silica a really high surface area. This means lots of space for polar molecules to stick to!
Page author: Rachel Chataway-Green