Download a printable version of this document here
Catalysts are useful tools in organic synthesis as they speed up a chemical reaction without being changed themselves, meaning they can be recovered at the end of the experiment and used again and again.
Catalysts speed up reactions by offering alternative reaction pathways. The energy needed for a reaction to occur is called the activation energy. If this is very high, the reaction requires high temperatures and can be very slow. Take a look at the energy diagram here and think of it as the reaction needing to climb a very steep hill. This is very hard work and the reaction needs to use more energy to get over the top and reach the other side. This is slow work and it can be quite expensive to heat the system sufficiently to give it enough energy.
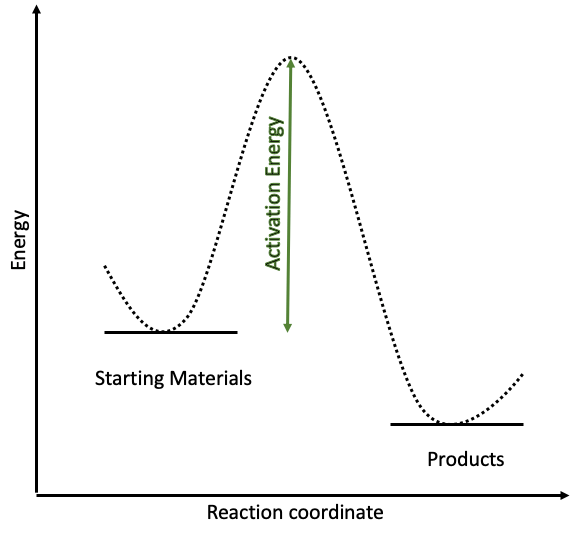
However, if we add a catalyst to this mixture, we can reduce the activation energy needed to complete the reaction.

The catalysed reaction in red has a much lower activation energy than the uncatalysed reaction. Going back to the hill from earlier, adding a catalyst into the mix makes the hike much easier, reducing the height of the hill and requiring less energy. This is great news for chemists because their reactions can happen much quicker under less severe conditions.
The best part is, catalysts are not directly involved in the reaction. As a result, you can reform/recover a catalyst at the end of an experiment, meaning we can keep using them for many different experiments.
Some reagents are sometimes mistakenly labelled as catalysts. An example of this is AlCl₃ which is used in Friedal-Crafts acylation. learn more on this chemBAM webpage; Is that a catalyst – Friedal-Crafts acylation.
Learn more about designing sustainable synthesis here.
Learn more about the chemistry of catalysis here.

This work is licensed under a Creative Commons Attribution 4.0 International License.