Download a printable version of this document here
Many materials are fluorescent, including the fluorescent highlighter pens that we use (look at these under a UV lamp!), our teeth, and a lot of white clothing. When something fluoresces, it absorbs light of a given wavelength (colour), and emits light and a different wavelength. For example, under black lighting in a disco, our teeth absorb UV light, and emit white light, so that they ‘glow’ in the dark. Ripe bananas fluoresce bright blue under similar UV light, due to one of the products of chlorophyll degradation.
Strictly speaking, the general process of absorbing light and re-emitting light of a different wavelength is called photo-luminescence.

During photo-luminescence, molecules absorb light and become ‘photo-excited’; some of the electrons within the molecule absorb the energy from the light and jump up to higher energy orbitals, to put the molecule into an excited state. Before the light is re-emitted from the molecule, it usually finds other ways to ‘relax’ to a lower energy state, and finally emits light (photons), to jump back down to the ground state. The wavelength of light that is absorbed and emitted corresponds to the energy gap that the molecule jumps across.
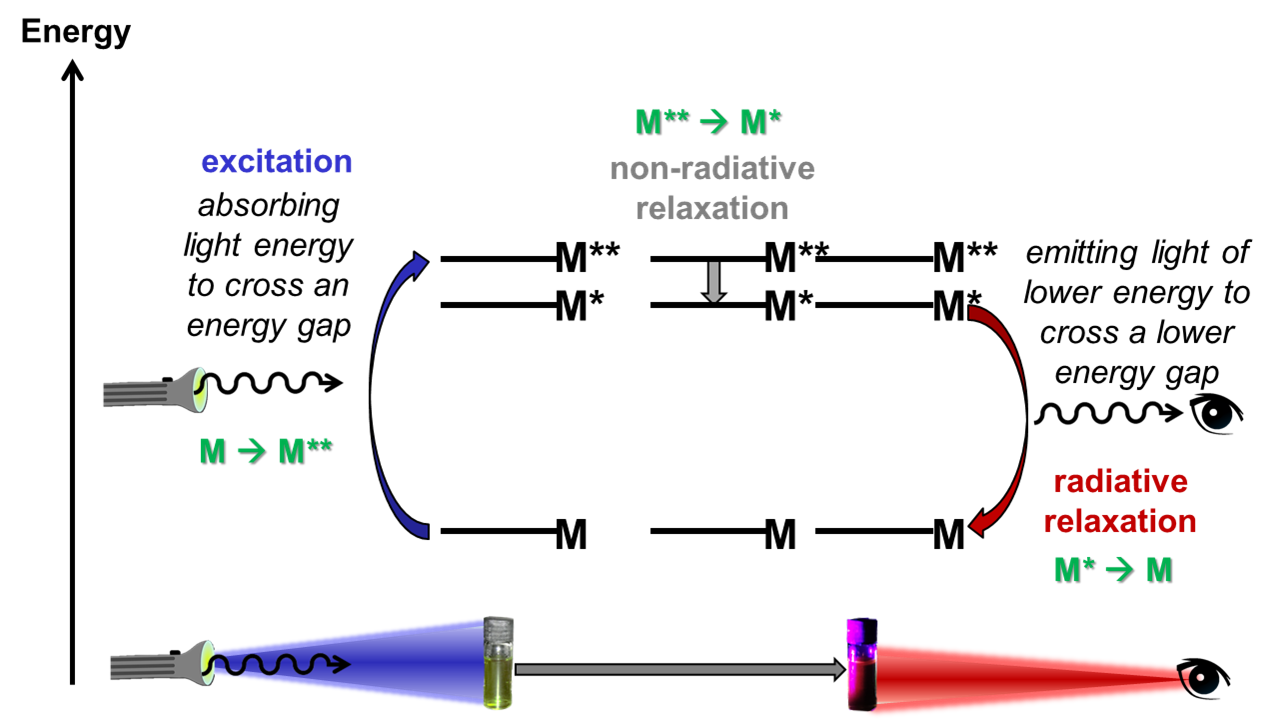

Fluorescence is one particular type of luminescence, in which this process occurs very quickly. After a molecule has absorbed energy from the light and has become excited, fluorescence occurs within a few nanoseconds (10-9 s); therefore you can only see the fluorescence when the excitation source (i.e. the UV Lamp) is switched on. The word fluorescence was coined in the 1850s, from the mineral fluorite, or fluorspar. Fluorite is a mineral form of CaF2 and many fluorites fluoresce due to the presence of impurities in the crystal structure.
Other types of luminescence, such as phosphorescence, occur much more slowly. Phosphorescent materials are used in ‘glow in the dark’ paint and stars; with these materials, they absorb light and become photo-excited, and then emit light long after the absorption process, and therefore the luminescence can be seen after the light has been switched off.
Luminescent materials have many applications, and thus their development is an ongoing area of research in science. We use fluorescent light bulbs in the home, optical brighteners in our washing powder, luminescent materials in plasma TV screens, and luminescent inks in bank notes, stamps, cheques, and driving licenses, as anti-counterfeit measures.

To learn more about sensing genetic disorders with fluorescence and more see the chemBAM general experiments page.

This work is licensed under a Creative Commons Attribution 4.0 International License.

