Download a printable version of this document here
Spectrometry and Spectroscopy are two terms which are very commonly confused. Both are types of techniques that we use to characterise molecules but they work in very different ways.
Spectroscopy
Spectroscopy is the study of how materials respond to electromagnetic radiation – that is, radiation with a wavelength from the electromagnetic spectrum.
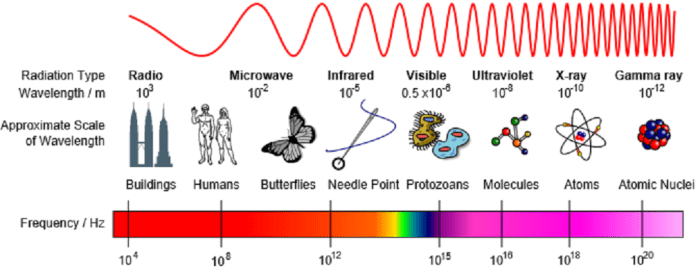
Examples include:
Ultra-Violet visible (UV-vis) adsorption spectroscopy; studies molecules which absorb UV-visible radiation. This is absorbed by valance electrons in a molecule.
Infrared (IR) absorption spectroscopy; studies molecules which absorb IR radiation. This can result in the bending and stretching of atomic bonds. This can help us to work out what functional groups are in a molecule.
Nuclear Magnetic Resonance (NMR) spectroscopy; studies how molecules respond to radio frequencies. In particular this causes some atomic nuclei to absorb energy and helps us to understand the environment they are in.
Spectrometry
Mass Spectrometry does not involve exposing a molecule to radiation. Instead it relies on high energy particles (such as electrons) to charge and fragment (break up) a molecule. The charged species are then separated according to their mass to charge ratio (m/z).

This work is licensed under a Creative Commons Attribution 4.0 International License.