Download a printable version of this document here.

Electrophilic substitution is a reaction which involves an electrophile reacting with a molecule with high electron density such as benzene. Benzene and its derivatives are commonly used in electrophilic substitution as benzene has 6 delocalised electrons forming its electron ring (also termed aromatic ring), therefore giving it a high electron density. This can also be visualised as 3 alkene bonds, both methods of visualisation illustrate how electrons are delocalised around the benzene ring.

A good example of electrophilic substitution reaction is the Friedel-Crafts acylation whereby an acyl group is added to an aromatic ring such as benzene. Here AlCl3 is required to form the reactive acyl intermediate (electrophile) to allow the electrophilic substitution to occur. To learn more about why AlCl3 is required and its role in the reaction see the chemBAM page; Is that a catalyst – Friedel-Crafts acylation.

A similar reaction is the Friedel-Crafts alkylation which involves adding an alkyl group to an aromatic ring such as benzene. This reaction was used commonly in the production of shampoos and surfactants.
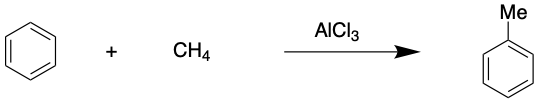
Task;
Using what you know, calculate how many tonnes of AlCl3 are required to react 10 000 tonnes of benzene to form methyl-benzene (aka toluene) if 1.05 moles of AlCl3 are required in the reaction?
Bonus task;
If you treat AlCl3 as a reagent in the reaction, can you calculate the atom economy and E factor of the reaction? Can you use this to justify why other “catalysts” were developed for this reaction?
For a recap on atom economy and E factor see the chemBAM page.
Download a version of this document with answers here.

This work is licensed under a Creative Commons Attribution 4.0 International License.