Background
Providing clean water for people to drink is one of the biggest challenges facing our world. Some countries struggle with water-borne diseases. Others face natural pollutants such as arsenic or man-made pollutants like textile dyes or pharmaceuticals.
Exercise: Get students to think of all the things that can cause water pollution. Download a full guide here.
One way to remove pollutants from water is to use activated carbon. This is carbon with lots of small holes, or pores, and it has a very high surface area. Activated carbon is normally made from biological materials such as wood, nut shells, peat or coal. If these are heated in an air-free environment (normally under nitrogen), they will turn into carbon. However, the carbon that is produced isn’t very porous and won’t adsorb pollutants.
To introduce pores into the carbon, the biological material can be heated to high temperatures in steam or oxygen. These react with the carbon to make carbon dioxide, burning away tiny holes in the carbon.
Once the carbon is full of small pores, it is very good at adsorbing organic molecules. These go into the pores and stick to the surface of the carbon. This is how activated carbon is used to remove pollutants from water.
Despite the range of activated carbons available, they can be too expensive for large-scale water treatment. Also, new types of porous carbons are needed. This is not just for water treatment, but for applications such as batteries or fuel cells.
Experiment
The ability of carbons to adsorb pollutants can be tested using dyes. The coloured organic molecules are adsorbed onto the carbon and this can be easily monitored by the loss of colour from a solution. In scientific research, a compound called methylene blue is often used. In this experiment we will use some fragrances, which are also organic molecules.
Aim
To investigate whether activated carbon or charcoal is better at removing an organic fragrance molecule from water.
You will need
- 3 palm sized pots with lids
- A beaker filled about halfway with tap water
- 3 centrifuge tubes or vials with lids, each labelled with a different shaped tactile dot
- 1 plastic funnel
- A small spoon
- Small bag of BBQ charcoal broken into small pieces (labelled with tactile dot to match one of the fragrance pots)
- Small bag of activated carbon (labelled with a tactile dot to match another of the fragrance pots)
Method
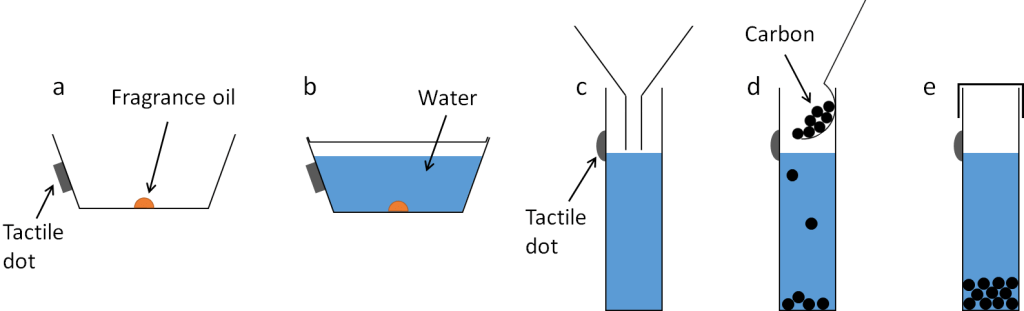
Instructions are available in the downloads at the top of the page, written below, or as a video below.
- Take three pots and put a drop of fragrance in each one. We tried wintergreen and cinnamon and they both worked well.
- Fill the pots almost to the top with tap water
- Close the lid carefully (you should hear it “click”) and shake gently to help the fragrance to dissolve in the water
- Use your small funnel to pour all of the solution from one pot into the centrifuge tube
- Repeat this with the other pots until you have three centrifuge tubes containing the fragrance solution
- Add two spoons of crushed BBQ charcoal to the first tube and put on the lid.
- Add two spoons of activated carbon to the second tube and put on the lid.
- Leave the third tube as it is with just the fragrance solution inside.
- Shake the tubes for approximately 30 seconds
- Leave for about 20 minutes and smell the pots.
Depending on the size of your spoon (we used a small measuring spoon) and where you buy your activated carbon from, you might need to use a different number of spoons of carbon.
Questions
- Why do we leave one tube with just the fragrance solution in it?
- Is there a significant difference between the fragrances when using activated carbon or charcoal?
- What happens when you leave the activated carbon in the fragrance solution for a longer period of time?
- What do you think would happen if you add more activated carbon or BBQ charcoal?
- Do you think using a different fragrance will have the same result, why?
- Where do you think this chemistry could be useful in the real world?
Discussion
In the experiment you used a porous carbon to remove fragrance molecules from water. The carbon is called porous because it has lots of little holes (pores) going through it.
When a material has lots of pores, it has a higher surface area.
When the carbon removes the pollutant molecules from water, the pollutant molecules stick to the surface of the carbon. This happens through interactions between the pollutant molecule (normally an organic molecule) and bonds on the surface of the carbon. If your carbon has a high surface area, there is more space for pollutant molecules to stick to.
Feeling porosity activity
Print out the shapes (see top of pwebpage) using swell paper or cut them out of cardboard and glue them to a piece of card or paper.
There are three shapes. The top shape represents a piece of carbon that has no pores. Run your finger around the edge. The second shape has some pores. The third shape has lots of pores. By running your finger around the edge can you determine which of the three shapes has the highest surface area?
Materials that have lots of small pores have the highest surface area. This can be hundreds of square metres per gram of material! A high surface area is good if you want to remove lots of pollutants from water.
A chemist’s perspective
Porous carbons are interesting because they can be made from natural materials like wood or nut shells. Many of these already contain some pores, although these are quite large. If we heat wood to a high temperature in a furnace without air, we can turn it into carbon.
There are also some clever ways that we can treat the carbons to get even more pores. This can include ‘burning’ extra pores into the carbon using steam or chemical compounds. This process is called activation. It is what is used to make activated carbons.