
Download a printable version of this document here
Retrosynthesis is a technique chemists use to identify how to synthesise a molecule through approaching the problem backwards. Effectively retrosynthesis starts from the wanted product and works backwards chopping and changing the molecule into smaller pieces to identify what the previous molecule can be made from. These “chopped up” molecules are referred to as synthons and are usually unstable ions, thus it’s important to identify a more stable equivalent molecule which would react in the same way. The trick to good retrosynthesis is knowing which bond to break, unfortunately this is something that comes with practice and experience!
What’s with the arrow?
A retrosynthetic arrow is used to show retrosynthesis is taking place through the breaking of a bond and NOT a reaction. The retrosynthesis arrow allows a clear distinction between a reaction taking place and retrosynthetic analysis.
Below are two examples of how retrosynthesis can be applied to two common prescription drugs, aspirin and paracetamol.
Aspirin
As shown in the first diagram, aspirin 1 can be chopped up into synthons 2 and 3. One question here is why break the bond in the position shown. The answer is if the bond is broken here the equivalent molecules 4 and 5 can react together through a simple nucleophilic addition-elimination reaction. But for instance molecule 5 (ethanoic anhydride) could be a range of electrophiles which react to add a acetyl group to a molecule.
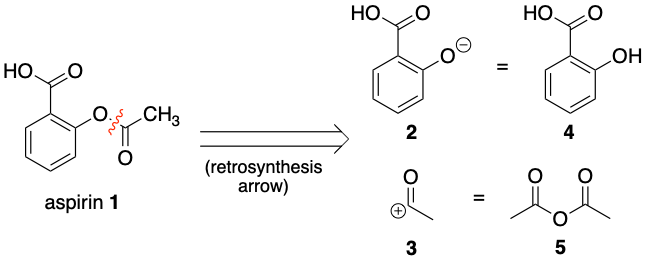
As shown in the second diagram, Salicylic acid 4 can be chopped up further into synthons 6 and 7. One big question here is why have the charges on the synthons that way around. The answer is if the charges are this way around, it is possible to react the equivalent molecules 8 and 9 together through electrophilic substitution. Also if the charges on synthons 6 and 7 were the other way around, it would make little sense chemically, as it is highly unlikely an electron rich benzene ring would act as an electrophile.

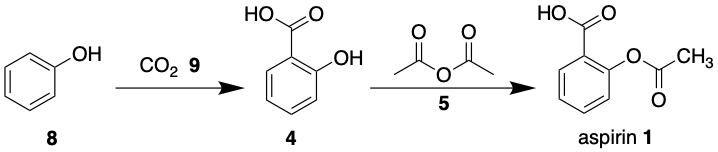
To summarise; aspirin can be taken back to common chemical starting materials, ethanoic anhydride 5, phenol 8 and CO2 9. Therefore the forward synthesis would be this;
Paracetamol
As shown in the first diagram, paracetamol A can be chopped up into synthons B and C. One question here is why break the bond in the position shown. The answer is if the bond is broken here the equivalent molecules Dand E can react together through a simple nucleophilic addition-elimination reaction as before.
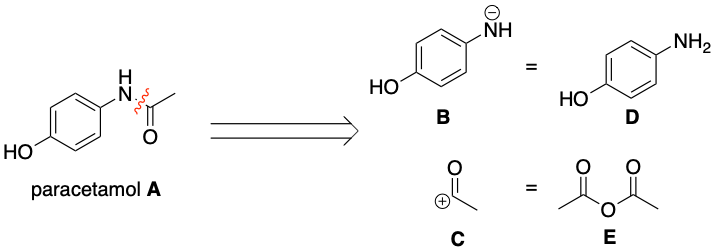
As shown in the second diagram, molecule D can be chopped up further into synthons F and G. One big question here is why has NO2 appeared? The answer is that NH2 is a poor electrophile (it usually acts as a nucleophile!). Therefore an equivalent electrophile for NH2 would be NO2 which can then be reduced to NH2. Thus NO2 can react with phenol (molecule H) through electrophilic substitution.
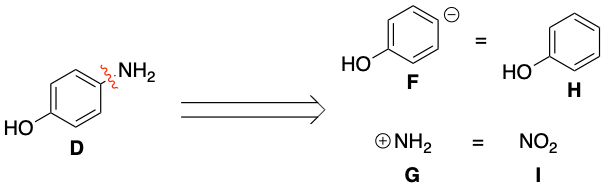
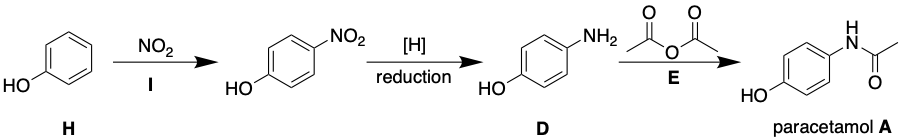
To summarise; paracetamol can be taken back to common chemical starting materials, ethanoic anhydride E, phenol H and NO2 I. Therefore the forward synthesis would be this;
Want to keep going? Try out this Aspirin Screen Experiment from the Royal Society of Chemistry.

This work is licensed under a Creative Commons Attribution 4.0 International License.