Download a printable version of this document here
Steady-state luminescence Spectroscopy
The emission spectra of fluorescent or luminescent solutions (see fluorescence) can be measured using a luminescence spectrometer, sometimes called a spectrofluorometer. This is an instrument that has a UV-visible excitation light source, often an xenon-arc lamp, to irradiate the sample, and the emission light is collected to form a spectrum:

The light from the xenon arc lamp (which contains wavelengths from 230-1000 nm, across a wide range of UV-visible-IR energies) passes through a monochomator, which diffracts the light and disperses it into different wavelengths, in a similar fashion to a prism. By changing the angle of the diffraction grating, a specific wavelength of light is selected, so that we can irradiated our sample with a known wavelength of light. This light excites the molecules in the solution of a sample, which we put in a transparent cuvette made of quartz (if we use glass, we cannot irradiate it with UV light, as glass absorbs light at shorter wavelengths than ~ 330 nm). The light emitted by the sample is collected perpendicular to the excitation, to reduce the excitation light being detected. The light is passed through another monochromator, and the intensity of light at each wavelength is recorded, as the monochromator rotates and scans through a range of wavelengths (relating to a range of angles of the grating), to create a spectrum. The visible light is collected by a photomultiplier tube (PMT), which detects photons (light) and converts this into a voltage, to record the signal electronically.
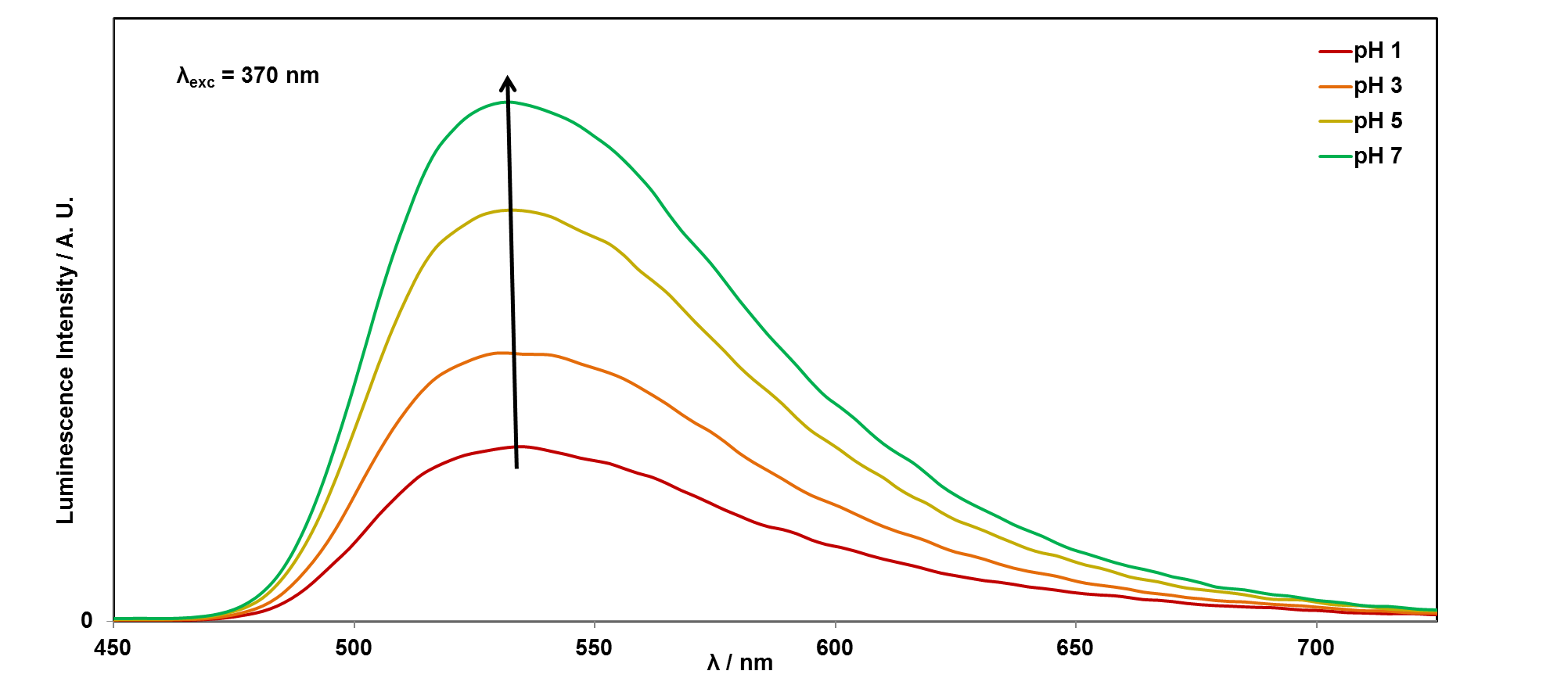
In this example, the cuvette of riboflavin, dissolved in water, was irradiated with the Xenon lamp set to 370 nm, and the emission (luminescence) signal was collected as the emission monochromator was scanned between 450-725 nm. The most intense light from riboflavin is emitted at 530 nm, and hence it looks yellow/green under UV light.

This work is licensed under a Creative Commons Attribution 4.0 International License.