
Download a printable version of this document here
Isomers in general terms are molecules which have the same molecular formula (same number of atoms), but are arranged/displayed differently to give distinct structures.
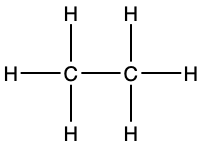
Molecular formula; shows the actual number of each type of atom in a molecule e.g. C2H6
Structural formula; shows all the covalent bonds present in a molecule in 2 dimensions e.g;
Structural isomer
Definition; structural isomers have the same molecular formula, but their atoms are arranged differently in space.
Structural isomers can be separated into 3 groups;
- Chain isomer; molecules with the same molecular formula, but different structures of the carbon skeleton in space.

- Position/regio isomer; molecules with the same molecular formula, but different structures in space due to different positions of the same functional group on the same carbon skeleton.

Functional group isomer; molecules with the same molecular formula, but with atoms arranged to give different functional groups in space.
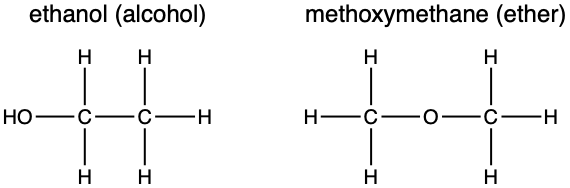
Stereoisomers
Definition; stereoisomers have the same structural formulae, but have a different arrangement of atoms in three dimensional space.
Stereoisomers can be separated into 2 groups;
- Geometrical (E-Z) isomers; occur due to restricted rotation around a alkene (C=C) double bond.
- E-Z stereoisomers arise when:
- There is restricted rotation around an alkene (C=C) double bond.
- There are 2 different functional groups/atoms attached both ends of the restricted double bond.

- Optical isomers; occur when there is a stereogenic centre in a molecule, i.e. an atom which is bonded to 4 different functional groups/atoms.
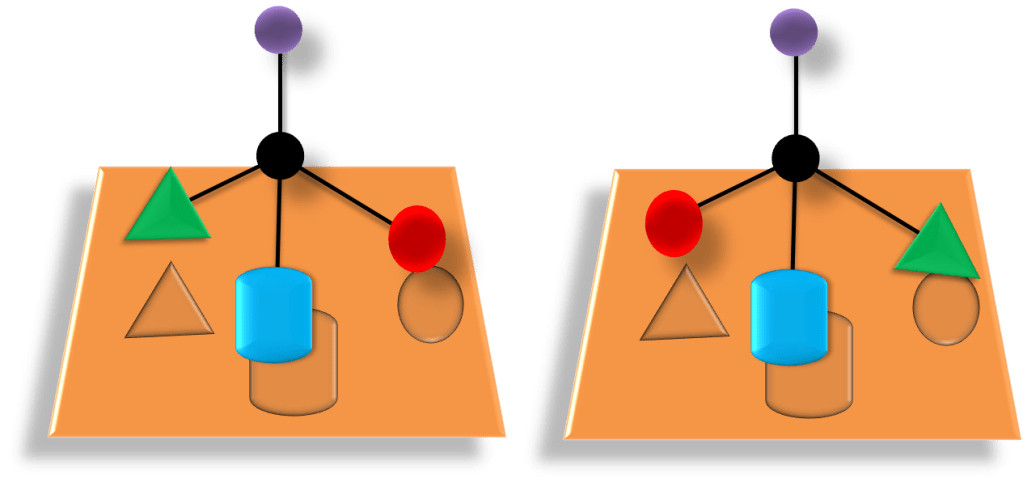
To learn more about optical stereoisomers; enantiomers and diastereomers see the chemBAM page.
To learn more about optical isomers in healthcare and a related experiment, see the chemBAM page.

This work is licensed under a Creative Commons Attribution 4.0 International License.