
Background
As chemists, we often need to separate and purify mixtures of molecules. When we synthesise new chemicals, we rarely make one pure product without side products or unreacted starting materials or catalsyts that are present in the mixture with the product. We don’t always make new compounds that need separating, sometimes we wish to extract naturally-occuring molecules from mixtures.
There are several ways to separate different chemicals from one another, and the purification of molecules is often the hardest part of a chemist’s job!
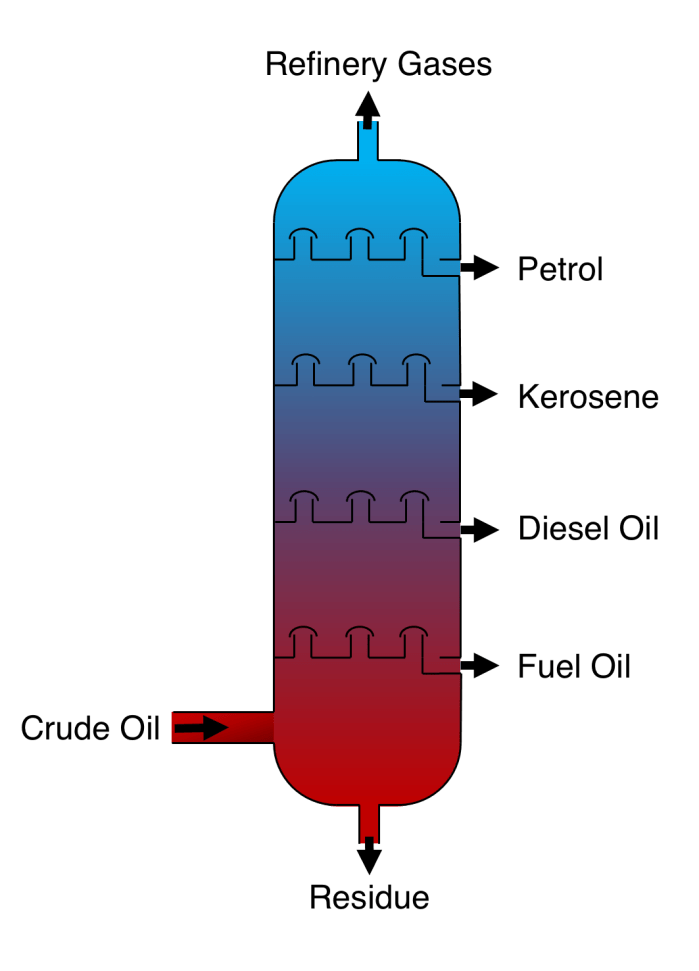
When we talk about fractional distillation we are normally discussing the fractional distillation of crude oil. Crude oil is the unrefined substance that we find in the earth. We can use the fact that the molecules we wish to separate have different boiling points. Molecules have different boiling points for many reasons but in this case it is mainly due to the different length of the molecules. The smaller molecules have a lower boiling point whereas the larger molecules have a higher boiling point. In industry a tall column is used to distill the mixture with condensers at different heights. The mixture is heated and substances with a low boiling point will condense at the top of the column whereas substances with a high boiling point condense at the bottom of the column. When it undergoes fractional distillation crude oil separates into many useful compounds, including refinery gases, petrol, kerosene, diesel oil, fuel oil, and a residue (as depicted in the image below).
Crude oil contains water, salts and sulphur that need to be removed before the crude oil can be refined to its useful components. To remove the salt and the water, crude oil undergoes a step known as desalting. In this step the crude oil undergoes an initial dilution with the addition of clean water. The new diluted mixture is then emulsified so that the salty water initially present in the crude oil comes into contact with the clean water. The emulsion then enters the desalter which separates the emulsion into two separate layers, a layer of water and a layer of crude oil. Salt easily dissolves in water and because of this it is also separated along with the water. The water is then sent to the water treatment system and the crude oil progresses to the next stage. The next stage is to desulfur the crude oil, this is important as this should reduce the emission of sulfur dioxide (SO2) when any fuel created in the fractional distillation is burnt. Desulfurisation requires heating and pressurising the crude oil to specific conditions and then the addition of hydrogen gas. The addition of the hydrogen gas should cause a reaction with the any sulfur present and form hydrogen sulfide (H2S). The hydrogen sulfide is then converted into elemental sulfur and hydrogen, the sulfur can be sold on as a by-product whereas the hydrogen can be recycled and used again in the previous step.
Water treatment is very important in an oil refinery as water is used or removed in most parts of the plant. Initially oil skimmers are used, which are pieces of equipment that remove any oil that would be floating on the surface of the water. The next stage incorporates several large tanks that force air through the water. The water is mixed with biological agents that will consume any unwanted waste in the water and the air that is forced into the water helps promote the growth of these agents. To find out more about water treatment click here.
Experiment
A downloadable version of this experiment is available here. Please ensure you refer to the safety card. Details for teachers or technicians can be found here.
AIM
To separate a fizzy drink into its ingredients by fractional distillation.

YOU WILL NEED
- Conical flasks (at least 100 ml)
- Tripod/stand and clamp
- 3 Test tubes/vials per group
- Test tube holder
- Bunsen burner/heat source
- Fizzy drinks (worked well with pink Lucozade, and various flavours of Fanta)
- Universal indicator
- Ice
- Large beaker/container to hold ice
- Rubber bung with two holes
- Plastic/ Teflon tubing
- Thermometers
PROCEDURE
Part 1: release of CO2
Measure out 75 mL of the soft drink into a conical flask.Fill a test tube/vial ¾ of the way full with tap water. Add 1 mL of universal indicator and note the colour and pH of the tap water. Place the conical flask on top of the tripod or clamp it securely. Push the thermometer through one of the holes in the bung and the tubing through the other. If there is an issue with getting the thermometer or tubing through the bung add a bit of washing up liquid to the the end that you want through the bung. The thermometer should rest above the liquid and measure the temperature of the vapour. Carefully fit the bung on top of the conical flask making sure that there is a tight seal, and put the other end of the tube in the universal indicator solution. Place the Bunsen burner/heat source under the conical flask. Gently heat until the thermometer reads around thirty degrees, or there is a consistent release of gas. As the gas passes through the universal indicator solution in the test tube/vial note the colour change and the pH.
Part 2: distillation of odorous compounds
Fill a beaker or container with ice, and place a second test tube/vial in the ice. Swap the tube that is running into the first test tube an place it in the second test tube. Gently increase the heat of the soft drink until it begins to boil and produce a distillate. Collect the distillate up to about 20 mL then remove the test tube/vial from the beaker/container.
Part 3: distillation of the water
Place a third test tube/vial in the beaker/container of ice, and swap the tubing from the second test tube to the third test tube/vial. Maintain the heat on the soft drink and collect the remaining distillate. Observe what remains in the conical flask. Compare the smell of test tube 2 and test tube 3.

This work is licensed under a Creative Commons Attribution 4.0 International License.