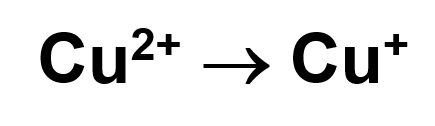Proteins
Proteins are essential molecules of all organisms – they co-create the wall of cells and take part in vast majority of biochemical reactions inside the human body. The main body of a protein consists of a peptide chain made up of amino acids (more precisely α-L-amino acids) bonded together by peptide bonds.
Amino acids are organic acids with at least one amine (–NH2) group and carboxyl (–COOH) group. There are thousands of amino acids that have been discovered, however, only 20+2 of those are found in proteins. The 20 acids are referred to as encoded (proteinogenic) amino acids and the 2 are special kind of acids. More on amino acids and proteins can be found here.
Saccharides
Saccharides are an important part of nutrition. Their molecules are made up of monosaccharide units and are present in all organisms. Water-soluble monosaccharides and oligosaccharides have sweet taste and are called sugars.
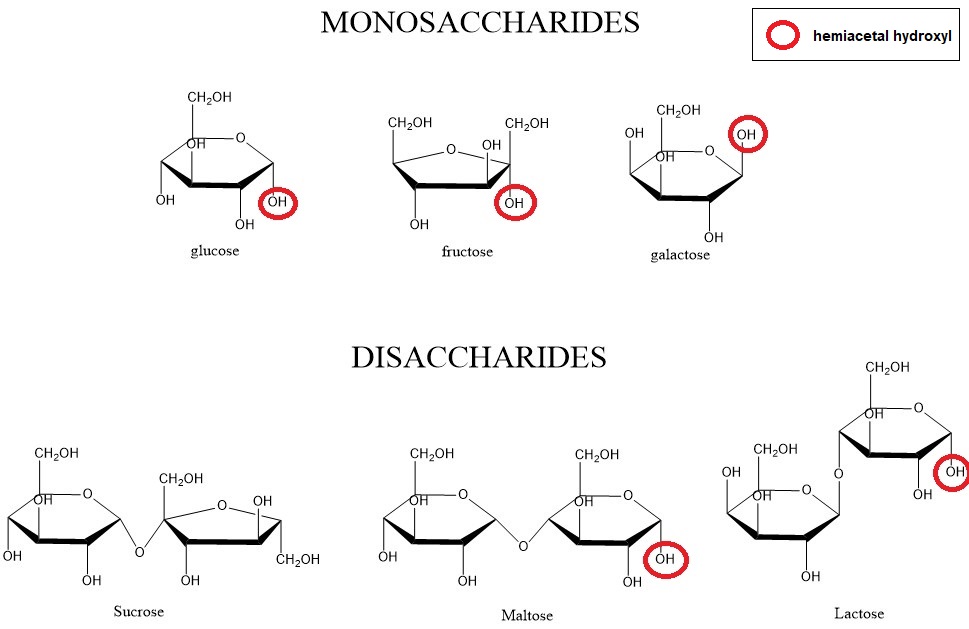
We can classify them based on the functional group present in the molecule as either aldoses or ketoses. Aldoses are derived from the simplest monosaccharide glyceraldehyde and they contain an aldehyde (–COH) functional group. Ketoses are derived from dihydroxyacetone and contain a ketone (–C(O)–) functional group.
Hemiacetal bond can be generated between the carbonyl (ketone/aldehyde) carbon and the hydroxyl (–OH) group on the 4th or 5th carbon atom in aqueous environment. Therefore we get a cyclic form (called Haworth projection) of saccharides. Hemiacetal hydroxyl group has different chemical properties from other hydroxyl
groups within the molecule – it can easily react with another saccharide and form a disaccharide through the formation of glycosidic bond (–O–). With the formation of glycosidic bond, the cyclisation is no longer a reversible process.
More on saccharides can be found here.
Detecting Proteins – Biuret test
Proteins can be detected by so-called Biuret test. Biuret test shows us if the sample contains molecules with peptide bonds (i.e. proteins) by changing its colour to purple. The purple colour arises from a complex that is formed between copper ions and the peptide bond.
We use Fehling’s solution for this test. Fehling’s solution is prepared by combining two separate solutions – Fehling’s A and Fehling’s B. Fehling’s A is an aqueous solution of pentahydrate copper(II) sulfate (CuSO4.5H2O) and it is blue coloured. Fehling’s B is a colourless solution of aqueous potassium sodium tartrate (Rochelle salt) and potassium hydroxide (KOH) or sodium hydroxide (NaOH) in distilled water.
Detecting Saccharides
If one of the hemiacetal hydroxyls in the molecule is “free” (not modified by glycosidic bond), the saccharide can be oxidised by copper ions (Cu2+) or silver ions (Ag+). The ions undergo reduction during the reaction and so we call these types of saccharides reducing. Maltose, lactose and all monosaccharides are examples of reducing sugars. On the other hand, sucrose is not a reducing sugar as it doesn’t have a free hemiacetal hydroxyl.
To determine if samples contain reducing sugars, it is necessary to heat the samples (already in Fehling’s solution) over a flame. If the sample changes its colour to orange or red, the reaction is positive and the sample contains a reducing sugar. The colour change occurs due to the copper ions being reduced:
Copper ions in oxidation state 2+ are blue coloured, while in ox. state 1+ are orange or red coloured.
The experiment
Aim: to determine if food samples contain proteins and saccharides
You will need:
- 9x test tubes
- Test tube rack
- Plastic pipette
- Distilled water
- 8x beaker
- Glass rod
- Test tube clamp
- Burner
- Fehling’s A (aqueous solution of pentahydrate copper(II) sulfate)
- Fehling’s B (solution of aqueous potassium sodium tartrate and potassium hydroxide or sodium hydroxide in distilled water)
- 8x different food samples
- Based on chosen food might also need knife, strainer or blender (mixer)
Procedure
Firstly prepare your samples. If you are working with liquid samples (e.g. some drinks), you can pour those into beakers straight away (don’t forget to mark your beakers!). If you are working with solid samples (e.g. sausage) use blender, add distilled water and filter through strainer before pouring it into beakers.
Add a bit of your samples into test tubes (about 2 cm). Add 2 ml Fehling’s B and then 2 ml Fehling’s A to your samples in test tubes and shake them carefully. Make also a blank test – add both Fehling’s into a test tube containing only distilled water. You can use the blank test for comparing colours. Write down your findings into the required section of the worksheet.
One by one, heat the test tubes over a burner. Be careful as overheating could lead to the sample spraying out of the test tube. Make sure you point the test tube away from yourself and anyone around you! Fill in the required section of the worksheet.
Results
We decided to test 13 samples – milk, white yogurt, egg yolk, egg white, sausage, Ladyfingers (biscuits), cucumber, potato, apple, orange, raisins, Vinea drink, Sprite zero.
First, we juiced the orange (squeezed out its juice) and filtered it through strainer so there were no seeds in our sample. As for sausages, we chopped them and used blender to mix them into a mash with the addition of a little bit of distilled water. Once they were mashed thoroughly, we filtered the solution using a strainer. Cucumber was sliced, mixed and filtered and the same was done with the potato and apple. Ladyfingers were soaked into distilled water, mixed with blender and filtered too. Raisins were cooked with water, mixed and then filtered. A bit of distilled water was added to white yogurt so it could be handled more easily. Lastly, egg yolk and egg white were diluted a bit with distilled water.
Our results were as follows:
With the addition of Fehling’s solution into our samples, some changed their colour to purple meaning the copper ions formed complexes with peptide bonds. This means that the Biuret test was positive and those samples contain proteins. Those samples that were blue-coloured don’t contain proteins – the blue colour is due to the Fehling’s solution being blue on its own.
Upon heating the samples, those that changed their colour to orange demonstrate the presence of reducing sugars. It is important to note that this experiment detects only reducing sugars (so if a sample contains sucrose, the test is negative).