Proteins are essential molecules of all organisms – they co-create the wall of cells and take part in vast majority of biochemical reactions inside the human body. The main body of a protein consists of a peptide chain made up of amino acids (more precisely α-L-amino acids).
Amino acids
Amino acids are organic acids with at least one amine (–NH2) group and carboxyl (–COOH) group. There are thousands of amino acids that have been discovered, however, only 20+2 of those are found in proteins. The 20 acids are referred to as encoded (proteinogenic) amino acids and the 2 are special kind of acids.
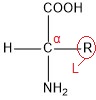
All proteinogenic amino acids, with the exception of Proline, belong among α-amino acids. The “α-“ in the name means that the amine group is bonded to carbon in alpha position to the carboxyl group. Furthermore, all proteinogenic amino acids, except Glycine, possess stereogenic centre and have L-configuration on the carbon adjacent to the carboxyl group. The letter “L-“ relates to stereochemistry of the compound and tells us about the relative configuration of the molecule, compared to the enantiomers of glyceraldehyde as the standard compound.
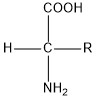
A general formula for encoded amino acids is:
“R” stands for either hydrogen atom or a carbon chain radical. The chain (or H atom) is not part of the peptide chain but forms a protrusion from the main chain; for this reason it is referred to as the side chain of an amino acid. Based on the type of the side chain, we classify amino acids as apolar, polar neutral, acidic or basic. Apolar acids contain only apolar side chains (e.g. alkyl groups). Polar neutral have non-ionizable polar group in the place of the side chain; they don’t have a charge in aqueous solution but the polar group easily interacts with water and hence increases the miscibility of the amino acid. Acidic amino acids have a negative charge on the side chain in neutral environment while basic acids have a positive charge.
Names of amino acids in peptides are abbreviated into 3 letters; this naming system is international.
Amino acids can be characterised based on their importance for human nutrition. Our organism cannot synthetize some amino acids and we need to receive them from the food – these amino acids are referred to as essential (or indispensable). The rest of the amino acids are called nonessential. Dietetics (science of nutrition) distinguishes proteins as either complete (contain all essential amino acids) or incomplete (contain only a few essential acids or none). Animal proteins (e.g. meat, fish, eggs, dairy) are often complete while plant-based proteins (e.g. rice, beans) are incomplete.
| Name | Abbreviation | Formula | Nonpolar/ Polar neutral/ Acidic/Basic | Essential/ Nonessential |
| Glycine | gly |  | N | N |
| Alanine | ala | 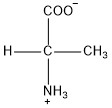 | N | N |
| Valine | val | 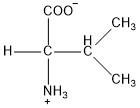 | N | E |
| Leucine | leu |  | N | E |
| Isoleucine | ile | 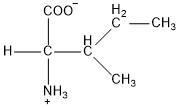 | N | E |
| Proline | pro | 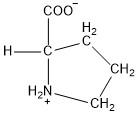 | N | N |
| Phenylalanine | phe | 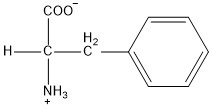 | N | E |
| Tyrosine | tyr | 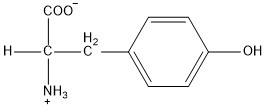 | P | N |
| Tryptophan | trp | 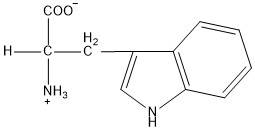 | N | E |
| Histidine | his | 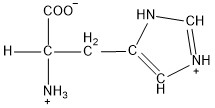 | B | N |
| Serine | ser | 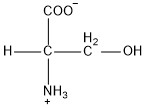 | P | N |
| Threonine | thr | 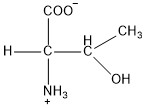 | P | E |
| Cysteine | cys |  | P | N |
| Methionine | met | 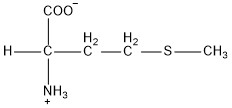 | N | E |
| Lysine | lys |  | B | E |
| Arginine | arg | 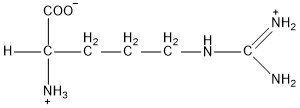 | B | N |
| Aspartic acid | asp | 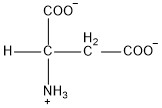 | A | N |
| Glutamic acid | glu | 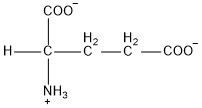 | A | N |
| Asparagine | asn | 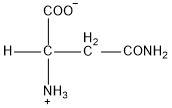 | P | N |
| Glutamine | gln |  | P | N |
Peptides
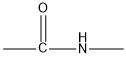
Two amino acids can react together and form a peptide (in this case dipeptide). In short, the amine-end (–NH2) of one acid reacts with the carboxyl-end (–COOH) of the other acid and forms a peptide by the formation of peptide bond.
Peptides can be classified into groups based on the number of amino acids they are made up of:
- Oligopeptides – 2 to 10 amino acids
- Polypeptides – 10 to 100 amino acids
- Proteins – more than 100 amino acids
General scheme of two amino acids bonding together:

Function of proteins
All biological systems are related to proteins with their life functions:
- Structural proteins (e.g. collagen, keratin) play structural function and are part of the primary building material of extracellular structures (e.g. cartilage, hair, nails)
- Transport proteins (e.g. haemoglobin, myoglobin) serve the function of moving other materials within an organism
- Storage proteins (e.g. ferritin) serve as biological reserves of metal ions and amino acids used by organisms
- Enzymes (e.g. lipases, amylases) are biological molecules that significantly speed up the rate of chemical reactions that take place within cells
- Antibodies or immunoglobulins (e.g. IgG, IgA) play a role in the immune system
- Contractile proteins (e.g. actin, myosin) are involved in muscle contraction and movement
- Hormonal proteins (e.g. insulin) function as chemical-signaling molecules that act to control or regulate specific physiological processes (e.g. growth, metabolism, reproduction)
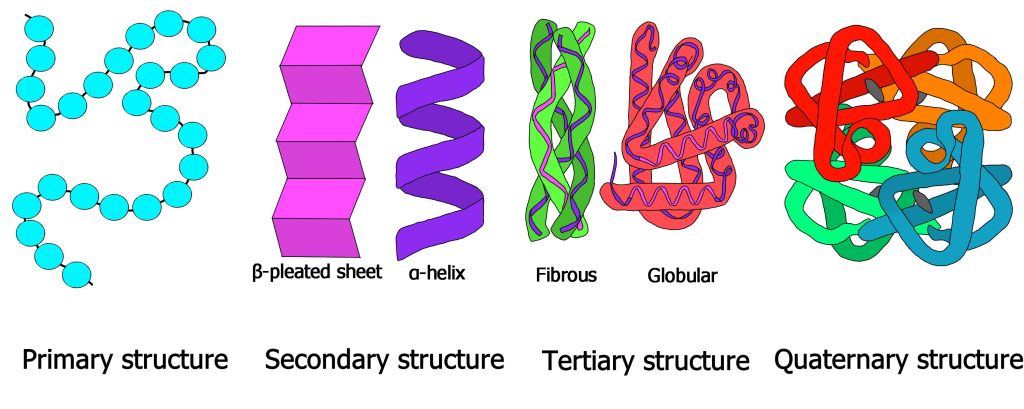
Structure of proteins
Proteins have unique precisely organised and effectively stabilized spatial structures. We can characterise the different structures as:
- Primary
- Secondary
- α-helices
- β-structures (also called β-sheet or β-pleated sheet)
- Tertiary
- Globular (or spheroproteins)
- Fibrous
- Quaternary
Primary structure is a chain of amino acids denoting the order of individual amino acids in the chain. This type of structure doesn’t contain the full information about the chemical structure. However, it does contain the fundamental information about the protein’s spatial structure.
Secondary structure determines the spatial (geometrical) structure of only a part of the chain. We distinguish α-helix and β-pleated sheet structures. The structures are reinforced by hydrogen-bonds (a bond between H and O).
Tertiary structure conveys the geometrical arrangement of the whole chain without the prosthetic group (a non-protein structure adjacent to the protein). Two types can be considered. Globular proteins have spherical (“globe-like”) shape while Fibrous proteins have an elongated shape.
Quaternary structure relays the geometrical arrangement of the whole chain including the prosthetic group.
For more information on protein structures visit here.
For a laboratory experiment on saccharides and proteins click here.
Links for download:
List of proteinogenic acids as pdf or jpg .
Worksheet on proteins & amino acids, together with answers can be found here.