Molecules of saccharides are made up of monosaccharide units and are present in all organisms. Based on the number of these units in the molecule, we classify saccharides as:
- Monosaccharides – 1 unit
- Oligosaccharides – 2 to 10 units
- Polysaccharides – more than 10 units
Water-soluble monosaccharides and oligosaccharides have sweet taste and are called sugars. Saccharides are an important part of nutrition.
Structure
Based on the number of carbon atoms within the molecule, we distinguish:
- Trioses – 3x C (e.g.glyceraldehyde, dihydroxyacetone)
- Tetroses – 4x C
- Pentoses – 5x C (e.g. ribose, ribulose)
- Hexoses – 6x C (e.g. glucose, fructose)
- Heptoses – 7x C
Furthermore, we differentiate between two main groups of saccharides based on their functional group in the molecule – aldoses and ketoses. Aldoses are derived from the simplest monosaccharide glyceraldehyde and they contain an aldehyde (–COH) functional group. Ketoses are derived from dihydroxyacetone and contain a ketone (–C(O)–) functional group.
Now try this “spot aldose/ketose” quiz. (opens as google forms)
Saccharides possess chiral centre(s), hence we get optical isomers (enantiomers) which are capable of rotating the plane-polarized light in different directions. Fisher nomenclature uses “D-“ and “L-“ prefixes to discriminate between the isomers. The “D-“ stands for “dextro” (meaning right) and signifies that the OH group on the penultimate carbon in the chain is on the right. The “L-“ stands for “levo” (meaning left) and conveys that the OH group on the penultimate carbon in the chain is on the left. In nature, we mostly see D-saccharides as these form a more stable cyclic form than L-saccharides. In fact, polysaccharides we get from food are made up of D-saccharides only. However, L-saccharides can also be found in nature and be metabolically processed by our bodies. This phenomenon is called homochirality (suggesting presence of only one isomer) and occurs for amino acids as well. In case of amino acids, L-amino acids are predominantly present in nature in proteins. The reason behind this homochirality has been a topic of debate among scientists.
Hemiacetal bond can be generated between the carbonyl (ketone/aldehyde) carbon and the hydroxyl (–OH) group on the 4th or 5th carbon atom in aqueous environment. Therefore we get a cyclic form (called Haworth projection) of saccharides which is lower in energy than the chain form and hence occurs predominantly in nature. In this form the molecule has increased number of chiral centres.

Cyclic isomers of saccharides are denoted as either “α-“ or “β-“. The cyclic forms remind us of pyran or furan and so when naming a cyclic saccharide we use these names (e.g. β-D-glucopyranose, α-D-fructofuranose). The backward and forward reactions of cyclisation are in equilibrium and so all three forms exist in equilibrium.

Hemiacetal hydroxyl group has different chemical properties from other hydroxyl groups within the molecule – it can easily react with another saccharide and form a disaccharide through the formation of glycosidic bond (–O–). With the formation of glycosidic bond, the cyclisation is no longer a reversible process.
Monosaccharides
The most important monosaccharides include glucose, galactose, fructose, ribose and deoxyribose.
D-Glucose is the most abundant natural monosaccharide. It can be found in blood where its concentration needs to be strictly regulated by the organism, otherwise it leads to diabetes. Plants and most algae produce glucose during photosynthesis from water and carbon dioxide, using energy from sunlight. Glucose is also present in cells in the form of ester with phosphoric acid (H3PO4) where it joins the metabolic system of the human body. Glycolysis (catalysed transformation of glucose) is one of the main sources of energy for cells; for this reason a solution of glucose is
used as artificial nutrition for patients.
Ribose and its derivate deoxyribose are the most important pentoses as they are part of nucleic acids.
Oligosaccharides and polysaccharides
Monosaccharides can be bonded together by glycosidic bond and form oligosaccharides or polysaccharides.
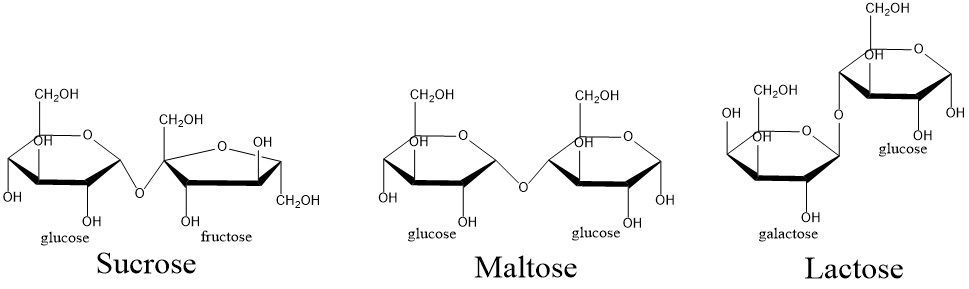
Sucrose is one of the most common disaccharides. It is made by combining glucose and fructose and plants use it for transporting saccharides from leaves to roots.
Maltose is a disaccharide formed from two units of glucose. It is the key building block of starch.
Lactose is composed of galactose and glucose subunits and is important for nutrition of all young mammals.
Starch is the most widespread storage polysaccharide formed by glucose units. It is the most common carbohydrate in human diets and is contained in large amounts in foods like potatoes, maize (corn) and rice. In industry, starch is converted into sugars and fermented to produce ethanol in the manufacture of beer, whisky and biofuel.
Cellulose is the main structural polysaccharide of plants and participates in creating the cell walls of microorganisms. Its molecules can be made up of up to 10 000 glucose units. In industry, cellulose is extracted from wood and used for producing paper. Cotton is essentially pure cellulose.
Functions of saccharides
Saccharides play several important functions in living organisms:
- Store energy (e.g. starch) and also take part in transfer of energy between organs (e.g. from liver to brain)
- Are main source of energy for chemotrophs (organisms that obtain energy by the oxidation of electron donors in their environments; e.g. animals, plants)
- Serve as a carbon-source for heterotrophs (organisms that cannot produce their own food; e.g. animals, fungi, bacteria)
- Are structural components of cells and tissue (e.g. cellulose, chitin)
- Are part of some proteins (glycoproteins) and of all nucleic acids (e.g. ribose, deoxyribose)
- Can be part of nucleotides and some vitamins
For a laboratory experiment on saccharides and proteins click here.
Worksheet on saccharides, together with answers can be downloaded here.



