You might have heard the word proteins before, probably when learning about the food pyramid, but what are they made of? Where do they come from?
We get a lot of protein from our diets and we need to make sure that we eat enough as it helps strengthen our muscles. In fact, a person weighing 90 kg should eat roughly 70 grams of protein a day! Foods like meat, eggs and nuts are all great sources of proteins but in fact all living things are made up of thousands of different proteins that all have specific jobs, such as adding structure to organs and tissues, or carrying molecules around the body.
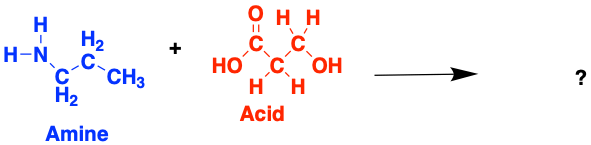
Proteins are made up of long chains of molecules called amino acids, named like this because they are made up of amines and acids. These are often referred to as the fundamental ‘building blocks’ of all proteins.
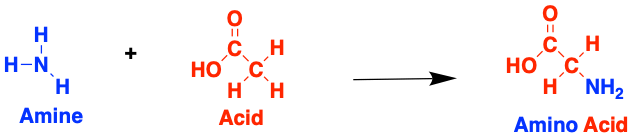
There are many different types of amino acid (this one above is called glycine) and they can combine in many possible ways to form tons of different proteins. When two amino acids join together they form a peptide, named after the bond that forms between them.
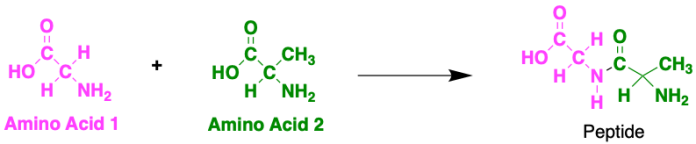
Proteins are made up of hundreds of thousands of these peptide-bonded amino acids in long chains! There are twenty possible amino acids in proteins, each with different side chains that provide different properties. As a result, the order of each of these in the chain can actually result in big changes in the properties of the protein itself. The order of the amino acids is determined by your DNA. DNA stores genetic information, which basically means it tells the body everything about itself, like eye colour, hair colour or whether you have dimples or not. It also tells your body which proteins to make depending on what you need.
The sequence of amino acids in the chain is known as the primary structure.
Exercise 1:
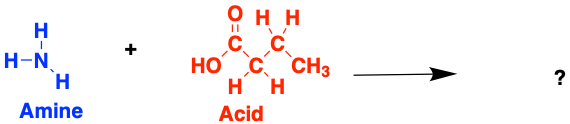
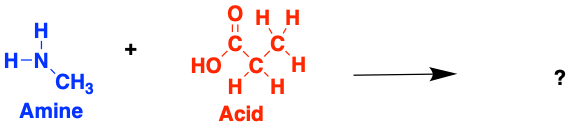
Have a go at building some (very small) proteins! Try and work out which amino acids form from each of these amines and acids. If you can, think about why the bonds are forming at the specific atoms you have chosen
1.
2.
3.
Now see if you can combine these three amino acids to form a peptide!
Extension:
Remember the order of amino acids in the chain is very important for the properties of the protein. Try and rearrange the order of amino acids to form a different peptide chain.
In real life, proteins aren’t just one really long straight chain. We also need to consider the secondary structure of a protein, which determines its shape. This is made up by alpha helices and beta sheets.
The amino acids in the peptide chain can rotate around their bonds making the protein very flexible. This allows the protein to fold in on itself. Hydrogen bonds can then form between different segments of the chain, keeping these folds in place. This is known as an alpha-helix as it gives the protein a corkscrew shape. Hydrogen bonds form between the acid group of one amino acid, and the amine group of another.
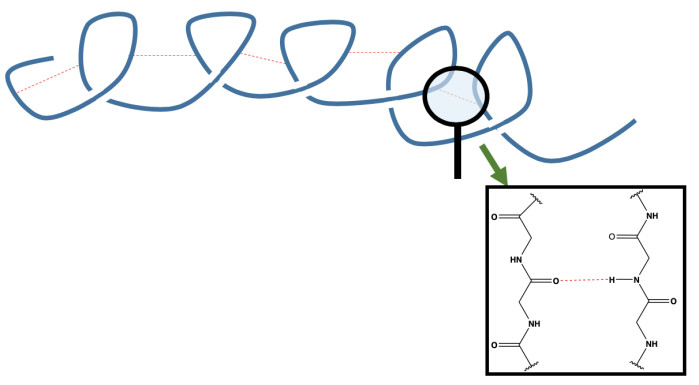
In this diagram of the alpha helix, we see hydrogen bonds (in red) holding together different sections of the protein chain, resulting in a spiral shape.
Another thing we need to remember is that proteins don’t exist as single chains, we get lots of protein chains that can interact with each other. We refer to these as beta-sheets which form when hydrogen bonds form between two different protein chains.
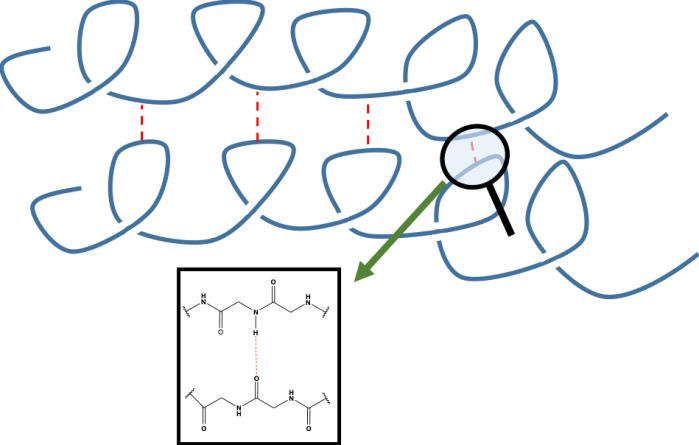
In this diagram we see two protein chains stabilised by hydrogen bonds (in red), again between the amine of one amino acid and the acid of another.
We also need to consider the interactions between different side chains on the amino acids in the protein. These can interact with each other causing the protein to fold even more and give it a 3D shape. These interactions are known as the tertiary structure. If the interactions between chains are lost, due to a breaking of hydrogen bonds for example, we see a loss in the shape of the protein. It is said to have been denatured. However, when the protein is denatured, the primary structure remains intact.
To learn more about protein structures (1°, 2°, 3°, 4°) see the chemBAM page.
Exercise 2:
Take a piece of string. This represents the primary structure of a protein. Wrap it around your hand in a helix (corkscrew) shape. When you remove it from your hand you will lose the shape of the protein.
However, if you add bag clips to the string, you can keep the alpha helix shape. These clips represent hydrogen bonds.
Now, with a partner, use more clips to join your polymer chains together forming the beta sheet structure.
Finally, with different coloured clips showing the different potential intermolecular bonds from the amino acid side chains, attach many different parts of the proteins together to form the 3D shape- the tertiary structure.
Now, with your complete structure, it is time to denature the protein. Undo all of the clips.
What happens to the primary, secondary and tertiary structure of a protein when it is denatured? What will we observe happening to the protein?
The Experiment
A pdf version of this experiment is available here. Please ensure you refer to the Safety card. Details for teachers or technicians can be found here.
AIM
To separate out the protein from a spirulina tablet and test the effect of different additives on the protein structure.
YOU WILL NEED
- 1 spirulina tablet
- 5 test tubes
- 150 mL of deionised water
- funnel
- filter paper
- torch (a phone torch is fine)
- Bunsen burner
- tripod
- 10 drops 0.5 M hydrochloric acid
- washing up liquid
- thermometer
- One egg
- Glass beaker
PROCEDURE
Part 1 – Spirulina
Crush a spirulina tablet into a fine powder and dissolve it in 150 mL of deionised water, stirring to help dissolution. This will take roughly 15 minutes
Filter to remove the undissolved chlorophyll until you get roughly 20 mL of the protein solution. Record any observations, e.g. changes in colour.

Add approximately 2 cm of the protein solution into each of 5 test tubes.
Using a torch, shine a light on the protein, recording the colour that you observe.
Leave one test tube as a control.
In test tube 2, add 2 drops of washing up liquid and shake. Note any colour changes. Shine the light on this solution, compare to test tube 1 and note any changes in fluorescence.
In test tube 3, add 5 drops of 0.1 M hydrochloric acid and shake. Note any colour changes. Shine the light on this solution, compare to test tube 1 and note any changes in fluorescence.
Now add another 5 drops of 0.1 M hydrochloric acid and shake. Note any colour changes. Shine the light on this solution, compare to test tube 1 and note any changes in fluorescence.
Finally, place test tube 4 in a water bath placed on a tripod. Start heating the water bath, keeping an eye on the temperature and the solution. For every 10 oC, note your observations of the protein solution, and any changes in fluorescence.
Once you notice a significant change in colour/fluorescence, take this test tube out of the water and leave to cool back to room temperature. Note any colour changes. Shine the light on this solution and note any changes in fluorescence. How does this compare to the heated sample?
At (or above) the same temperature that you removed test tube 4 from the water, heat test tube 5 for approximately 10 minutes. Note any colour changes. Shine the light on this solution and note any changes in fluorescence. Leave this to cool to room temperature and compare to test tube 4.
Part 2 – Egg white
Note: this part of the experiment doesn’t necessitate practical work and can just be discussed theoretically.
Crack open an egg and separate out the egg white from the yolk.
Put the egg white into a glass beaker above a Bunsen burner and heat it.
Note any changes that occur and think about what is happening to the protein structure.
Once you see a permanent change, remove the egg white from the heat and leave to cool to room temperature, or on ice. Note any changes in the protein structure of the egg white.
QUESTIONS
- As well as the protein, the spirulina tablets also contain chlorophyll. Why doesn’t this dissolve? What would happen if we tried to dissolve it in methanol?
- When you shine a torch on the protein what colour is it? What colour must the protein be absorbing? For more information on fluorescence take a look at the sensing with fluorescence experiment
- What happens to the fluorescence of the protein after heat/acid/soap?
- What do the observations in question 3 suggest has happened to the protein? Think about your answer in terms of molecular interactions.
- What happens to the heated protein in test tubes 4 and 5 when they return to room temperature? What conclusions can you draw from this?
- What happens to the protein structure of the egg white when you heat it? Does anything change when it is cooled? What can you determine from this?
IN THE RESEARCH LAB
Researchers working with Dr Anna Peacock at the University of Birmingham utilise proteins for many different purposes. In particular, their work involves designing peptide sequences capable of selective DNA recognition and binding, or as contrast agents in MRI.
Significant effort has been made into understanding how the protein matrix can affect the properties of the metal ion inside metalloproteins, with the ultimate goal being to generate artificial metalloproteins that are designed for specific purpose through tuning of the metal ion properties. One major way this is happening is the simultaneous de novo (from scratch) design of miniature artificial protein folds and metal ion binding sites. These metal ion binding sites can organise the peptide structure for functions such as enhanced binding to DNA.
Publications involving this research can be found below:
De Novo Design of Ln(III) Coiled Coils for Imaging Applications
Incorporating metals into de novo proteins
Files to download
pdf files:
editable files:
This work is licensed under a Creative Commons Attribution 4.0 International License.






